Arterial Switch
Overview
Arterial switch is a surgical procedure and the primary treatment for great artery transposition (TGA). The two major arteries leaving the heart are connected to the wrong ventricles, the two lower chambers of the heart, in transposition of the great arteries. As a result, oxygen-rich blood from the lungs is pumped back into the lungs, while oxygen-depleted blood, which is required to nourish the body, is pumped throughout the body.
Almost all TGA children have their arterial switches repaired. Babies with TGA become very ill soon after birth due to a severe lack of oxygen.
The arterial switch procedure is the primary treatment for correcting transposition of the great arteries (TGA). An arterial switch procedure is an open heart surgery that re-establishes normal blood flow in your baby's body. The aorta is detached from the right atrium and connected to the left atrium during surgery; the pulmonary artery is detached from the left atrium and tied to the right atrium; and the coronary arteries are cut out of the pulmonary artery and attached to the aorta.
What is Arterial switch?
The arterial switch operation (ASO) or arterial switch is an open heart surgical procedure used to correct dextro-transposition of the great arteries (d-TGA). It was developed by Canadian cardiac surgeon William Mustard and named after Brazilian cardiac surgeon Adib Jatene, who was the first to use it successfully. It was the first method of d-TGA repair to be attempted, but it was the last to be widely used due to technological limitations at the time of its development.
Historically, the arterial switch was preceded by two atrial switch methods: the Senning and Mustard procedures. The atrial switch, which attempted to correct the physiology of transposition, had serious flaws. The arterial switch attempted to address these issues. This has emerged as a physiologically and anatomically appropriate solution.
Background
One of the most significant achievements in pediatric cardiac surgery is the arterial switch operation (ASO). Before the first successful ASO, various palliative procedures, including atrial switches, were used to correct transposition of the great arteries (TGA)
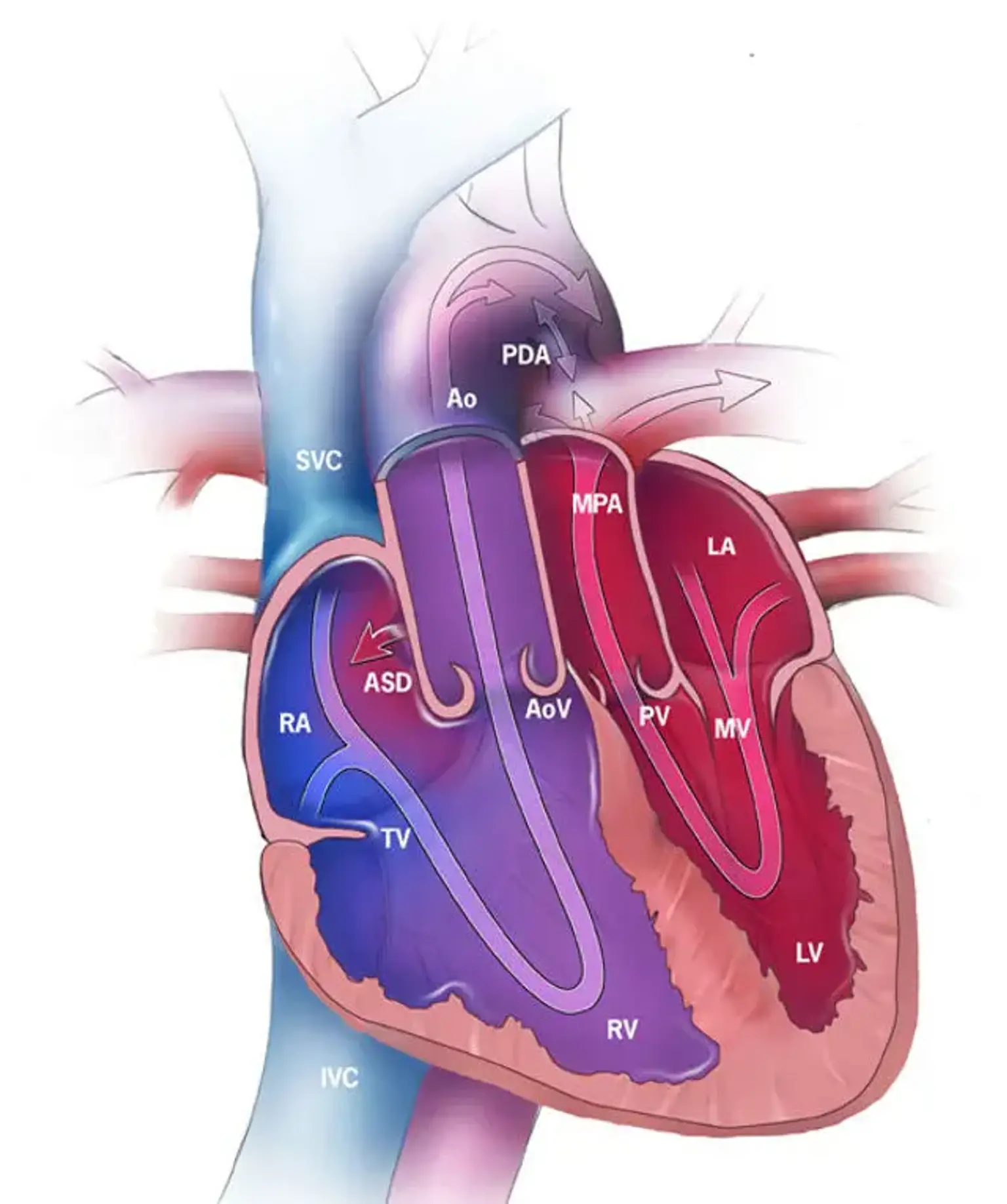
A heart with the two major arteries carrying blood away from the heart reversed. A normal blood pattern transports blood in the following order: body-heart-lungs-heart-body. When there is a transposition, the blood pathway is hampered because the two arteries connect to the wrong chambers of the heart.
This means that the blood flow cycle is stuck in either:
- Body–heart–body (without being routed to the lungs for oxygen) or
- Lungs–heart–lungs (without delivering oxygen to the body)
Without surgery, the only way to temporarily survive this condition is to have leaks that allow some oxygen-rich blood to cross into the oxygen-poor blood and be delivered to the body. A hospital can also catheterize a patient until corrective surgery is performed.
Permanent treatment necessitates a surgical procedure to realign the arteries. This procedure, known as an arterial switch operation, is performed within the first few weeks of life. It is an open-heart procedure in which the baby's heart is temporarily stopped while a heart-lung machine handles respiration and blood circulation. Any abnormal gaps between the ventricles or atria are also sealed.
The coronary arteries, which supply blood to the heart, must be removed from their normal position on the aorta and transplanted into the new "aorta," which now carries oxygenated blood from the left ventricle around the body. In rare cases, this may result in complications that necessitate additional procedures.
Despite early success, surgeons were hesitant to use the ASO instead of the well-established and far safer atrial switch procedure. In the 1980s, operative mortality for the ASO ranged from 30% to 60%, and it wasn't until the 1990s that most high-volume institutions achieved a low operative mortality rate.
What type of problems might my child have?
- Heart function problems
Patients who have undergone an atrial switch may experience a significant decline in heart muscle or heart valve function. This is due to the right ventricle pumping blood to the entire body rather than just the lungs. Medications that help the heart pump more efficiently, control fluid accumulation (diuretics), and control blood pressure may be beneficial. Patients who have undergone the arterial switch operation do not appear to be at as high a risk of heart muscle deterioration. However, they may have valve leakage or coronary artery problems.
- Heart rhythm problems (Arrhythmias)
People who have had their transposition repaired, particularly those who have had the Mustard or Senning operation, are at risk of developing heart rhythm abnormalities (arrhythmias). These arrhythmias frequently occur in the upper chambers of the heart. The heart rate of your child may be too slow or too fast. An artificial pacemaker can increase the heart rate if it is too slow. Medication can help slow your child's heart rate if it is too fast. Your child may require a cardiac catheterization to study and treat these rhythm disturbances at times.
What about surgical treatment?
There are two major types of surgery that can be used to correct the transposition. The first of these constructs a tunnel (a baffle) between the atria. This sends oxygen-rich blood to the right ventricle and aorta while sending oxygen-depleted blood to the left ventricle and pulmonary artery. This is referred to as an atrial or venous switch. It's also known as the Mustard or Senning procedure.
The arterial switch operation is the second type. The aorta and pulmonary artery are returned to their original positions. The aorta is linked to the left ventricle, while the pulmonary artery is linked to the right ventricle. The coronary arteries, which carry oxygen-rich blood to the heart muscle, must also be reconnected to the new aorta.
Surgical technique
The heart is accessed through a median sternotomy, and the patient is given heparin to prevent blood clotting. A large section of pericardium is harvested, disinfected and sterilized with a weak glutaraldehyde solution, and the coronary and great artery anatomy is examined.
To allow the vessels to move, the ductus arteriosus and right pulmonary branch, up to and including the first branches in the right lung's hilum, are separated from the surrounding supportive tissue. To indicate the commissure of the aorta to the neo-aorta, silk marking sutures may be placed in the pulmonary trunk at this time; alternatively, this may be done later in the procedure.
The cardiopulmonary bypass is then started by inserting a cannula as far away from the aortic root as possible while still supplying all arterial branches, another cannula is inserted into the right atrium, and a vent for the left ventricle is created via catheterization of the right superior pulmonary vein.
While the patient is cooling, the ductus arteriosus is ligated at both the aortic and pulmonary ostia before being transected at its center; the left pulmonary branch, including the first branches in the hilum of the left lung, is separated from the supportive tissue; and the aorta is marked at the site it will be transected, which is just below the pulmonary bifurcation and proximal to where the pulmonary artery will be transected.
When the patient has been completely cooled, the ascending aorta is clamped as close to the cannula as possible, and cardioplegia is achieved by delivering cold blood to the heart via the ascending aorta (below the cross clamp). The aorta is then cut at the marked location, and the pulmonary artery is cut a few millimetres below the bifurcation.
The vessels are checked again, and the pulmonary root is checked for left ventricular outflow tract obstruction (LVOTO). If a ventricular septal defect (VSD) is present, it can be repaired now via the aortic or pulmonary valve, or it can be repaired later in the procedure.
The great arteries are typically arranged using the LeCompte maneuver, with the aortic cross clamp positioned to hold the pulmonary artery anterior to the ascending aorta; however, with some congenital arrangements of the great arteries, such as side-by-side, this is not possible, and the arteries are transplanted in the non-anatomic 'anterior aorta' arrangement.
If the aortic commissure has not yet been marked, it can be done now using the same method as before the bypass; however, there is a third opportunity for this later in the procedure.
After the septal defects are repaired and the atrial incision is closed, the previously removed cannula is replaced and the HLM is restarted. The left ventricle is then vented, and the cross clamp from the aorta is removed, allowing full flow to be restored and rewarming to begin.
The previously harvested pericardium is then used to patch the coronary explantation sites as well as to extend - and widen if necessary - the neo-pulmonary root, allowing the pulmonary artery to be anastamosed without residual tension; the pulmonary artery is then transplanted to the neo-pulmonary root.
Preparation for arterial switch procedure
You will meet with a number of health-care team members prior to your newborn's surgery. The timing of these visits may vary, but the team will check in with you to ensure that you understand what is going on and that you have an opportunity to ask all of your questions.
Pre-operative tests
Before surgery all newborns have:
- Blood work
- An X-ray
- An echocardiogram
- An electrocardiogram
- An MRI imaging of the head
- All of these tests are done routinely to help the team prepare for surgery.
On the day of the procedure
On the morning of surgery, the anesthesiologist in charge will speak with you and answer any last-minute questions you may have. The operating room nurse will also greet you and perform a physical examination on your child. Parents usually do not accompany their children into the cardiac operating room for heart surgery for safety reasons. You will be asked to wait in a designated waiting room or another predetermined location.
During the procedure
Once your child is asleep, the team:
- Puts in place IV lines and other catheters
- Gives medications
- Cleans with antiseptic the area where the incision will be made
The anesthesiologist monitors your child and the monitors while communicating with other members of the surgery team to ensure that everything is stable during this open heart surgery.
Because your child requires a heart-lung machine, heparin (a blood thinner) is administered. The heart is stopped during this time while the surgeon repairs the defect. Your child is cared for by a perfusionist, anesthesiologist, and surgeon. They ensure that the heart-lung machine is delivering blood to the body and brain and injecting oxygen into the blood.
When the repair is finished, the heart is allowed to beat again, and the heart-lung machine is turned down and turned off. If the heart isn't working as well as it should, medications are used to improve heart function.
The cardiologist performs an echocardiogram to ensure that the repair is functioning properly. The surgeon then checks for bleeding and closes the incision. The majority of heart surgeries last between three and six hours.
After the procedure
The anesthesiologist will transfer your child to the cardiac critical care unit when:
- The team is pleased and confident that the repair has been completed successfully.
- The heart's function is normal and stable.
- Your child is thriving on the ventilator.
Recovery after the surgery
At hospital after the surgery
When your baby recovers from heart surgery, he or she will most likely spend a few days in the cardiac care until they feel better. Your child will then be transferred to the cardiac intensive care unit to complete their recovery. The cardiac team consists of:
- Ensures that your baby is not in pain by administering pain medication (most of the time acetaminophen; sometimes small doses of morphine as needed)
- Take care of your newborn's medications
- Manages the tubes (drains) that were implanted in your baby's chest during surgery and looks after the chest wound (sternal incision) and any other incision sites
- Ensures that your newborn can feed well by mouth.
Before going home
Your baby is ready to go home when they are breathing comfortably and can feed well by mouth. During the recovery, the cardiac team will teach you how to:
- Take care of the sternal incision
- Once at home, provide any medications that your baby may require and
- Attend to your newborn's basic needs, such as bathing, changing diapers, and safely holding your baby.
Ongoing Care
All patients with repaired transposition require routine care from an adult congenital heart disease specialist. EKG, echocardiogram, Holter monitor, and stress testing are all routine tests. Other tests, such as an MRI or a heart catheterization, may be recommended by your cardiologist. If you have a pacemaker, you will need to see the doctor more frequently. If you are undergoing any type of non-heart surgery or invasive procedure, you should also consult a cardiologist who specializes in caring for adults with congenital heart disease.
Medical Treatment and Follow-up
Medications that help the heart pump more efficiently, control fluid accumulation (diuretics), and control blood pressure may alleviate the symptoms of congestive heart failure. Some patients with extremely slow heart rates may require the use of a pacemaker. Those who have rapid heart rates will need medication to control them, or they may need a special cardiac catheterization (electrophysiology study) to study and treat these rhythm disturbances. Obstructions in the baffles are frequently treated with stents, which expand the obstructed regions.
Activity Restrictions
Many patients, particularly those who have undergone the arterial switch, will not need to limit their activity. It is generally recommended that patients undergoing atrial switch operations avoid activities that cause a rise in blood pressure, such as weight lifting. However, if your heart isn't pumping normally or if you have heart rhythm problems, you may need to limit your activity to endurance. Your cardiologist will advise you on whether you should limit your activity.
Pregnancy
Women who have had their transposition repaired may be able to have a successful pregnancy. If the mother has heart failure or arrhythmias, the risk of pregnancy increases. Before becoming pregnant, it is critical to consult with a cardiologist who is experienced in caring for patients with congenital heart defects to determine the health risks. It is also recommended that women with transposition be seen in centers that provide high-risk obstetrical services as well as adult congenital heart disease expertise.
Risks of an arterial switch procedure
Despite the fact that the arterial switch is a major surgical procedure, the outcomes are excellent. Ninety-nine percent of surgeries are completed successfully. This procedure will necessitate the use of cardiopulmonary bypass. Many children have minor issues, such as:
- Nausea
- Feeding problems (such as trouble swallowing)
- Bruising from intravenous (IV) sites
Rare, serious complications of anesthesia and heart surgery include:
- Bleeding, clotting, strokes
- Vocal cord injury
- Drug reactions
- Heart rhythm problems
- Cardiac arrest
- Reactions to a blood transfusion
- Brain injury
- Breathing problems
Prognosis
Early results from the arterial switch operation are excellent. Although the majority of patients grow into healthy adults, a small subset requires reoperation for pulmonary stenosis, neoaortic root dilatation, and neoaortic valve regurgitation, particularly as follow-up increases.
Patients with abnormal coronary anatomy, concomitant aortic arch obstruction, Taussig-Bing anomaly, ventricular septal defect, and low operative weight, as well as patients with an intact interventricular septum who present late, are challenging subgroups.
FAQ
1. Will my child need more surgery?
Some patients require additional surgery to help their hearts pump more efficiently, repair abnormal valves, or control heart rhythm disturbances. Patients who have had the Mustard or Senning operation may require surgery to correct atria tunnel abnormalities, repair abnormal valves, or control rhythm disturbances.
Patients who underwent arterial switch surgery may require additional surgery to relieve narrowings in the aorta or pulmonary artery where the original surgery was performed, or to repair leaky valves.
2. Will my child's activities be limited?
Most cardiologists advise patients to limit their physical activities to those that will test their endurance. They do not advise high school and college students to participate in competitive sports. Your child's cardiologist will assist you in determining the appropriate level of activity restriction.
3. What will my child need in the future?
Patients with transposition will need to see a cardiologist who is trained to care for patients with congenital heart disease for the rest of their lives. Your child may need to take medications to improve the function of his or her heart. A variety of non-invasive tests will be performed on your child by the cardiologist. Electrocardiograms, Holter monitors, exercise stress tests, and echocardiograms are examples of these tests.
4. What about preventing endocarditis?
Endocarditis prophylaxis is required for people who have had atrial switch operations and have had transposition of the great arteries. Most patients who have had simple arterial switch procedures will not require prophylaxis. Your cardiologist will be able to determine whether you need to continue taking routine antibiotics prior to certain dental procedures.
Conclusion
Complete transposition of the great vessels is the most common cause of death due to hypoxia (in the absence of an intracardiac shunt) or progressive heart failure.
The arterial switch procedure, also known as the Jatene switch procedure, is a surgical procedure used to correct transposition of the great arteries (TGA) at the aorta and main pulmonary artery.
Arterial switch procedures were created to address issues with earlier atrial switch procedures, such as the Senning or Mustard repairs. Atrial switch procedures have been linked to the late development of atrial arrhythmias, eventual right ventricular failure, and tricuspid regurgitation, as well as, in rare cases, pulmonary hypertension and baffle obstruction and/or leaking.