Glioblastoma
Overview
Glioblastoma, also known as glioblastoma multiforme (GBM), is the most malignant type of brain cancer. Glioblastoma symptoms and signs are initially nonspecific. They may include headaches, personality changes, nausea, and stroke-like symptoms. Symptoms frequently increase quickly and can lead to coma.
The majority of glioblastoma instances have no known cause. Genetic disorders such as neurofibromatosis and Li–Fraumeni syndrome, as well as past radiation therapy, are uncommon risk factors. Glioblastomas account for 15% of all brain tumors. They can arise from normal brain cells or from a pre-existing low-grade astrocytoma. A CT scan, an MRI scan, and a tissue biopsy are commonly used to make the diagnosis.
There is no known way to prevent the cancer. Surgery is usually the first step in treatment, followed by chemotherapy and radiation therapy. Temozolomide is a chemotherapeutic drug that is regularly used. To assist minimize swelling and discomfort, high-dose steroids may be utilized. Surgical excision of the tumor (decompression) is associated with enhanced survival, although only by a few months.
Despite the best treatment, cancer almost always returns. Following diagnosis, the average survival time is 12–15 months, with only 3–7% of persons living for more than five years. Survival without treatment is usually three months. It is the most prevalent type of brain cancer and the second most frequent type of brain tumor after meningioma.
Is Glioblastoma common?
The average yearly age-adjusted incidence rate (IR) of GBM is 3.19/100,000 population, according to the 2013 CBTRUS (Central Brain Tumour Registry of the United States) report. This is the greatest rate of occurrence for malignant brain and CNS cancers. GBM is most commonly diagnosed in adults, with a median age of 64. It is extremely uncommon among children. The incidence rises with age, with a peak between the ages of 75 and 84 and a decline beyond the age of 85. Given the aging population in the United States, the number of cases is likely to rise.
GBMs are more common in men; men had a 1.57 percent higher incidence rate than women. Primary GBMs are more common in men, while secondary GBMs are more common in women. GBM is more common in whites, followed by blacks. GBMs are most typically encountered in the supratentorial region, while they are extremely rare in the cerebellum and spinal cord.
What causes Glioblastoma?
Many genetic and environmental factors in glioblastoma multiforme have been explored, but no risk factor that accounts for a significant proportion of GBM has been identified. As with many other malignancies, GBM is sporadic, albeit one study found a high rate (17%) of past therapeutic irradiation among GBM patients. The time between irradiation and the onset of GBM ranges from a few years to several decades. Various studies linked the risk of GBM to lower susceptibility to allergies, immunological factors, immune genes, and some single nucleotide polymorphisms discovered in genome-wide association studies.
Gliomas are associated with a low risk of allergies and atopic illnesses, according to researchers. In addition, use of anti-inflammatory drugs for less than 10 years is related with a protective benefit against GBM in the near term. There is no convincing evidence that GBM is linked to lifestyle variables like as smoking, alcohol intake, drug use, or exposure to N-nitroso chemicals. The use of mobile phones does not raise the chance of developing GBM, according to studies; nevertheless, the relationship with long-term use requires more evidence.
How Glioblastoma develops?
Mutations cause aberrant proliferation, growth, and angiogenesis in malignant cells. Many genetic and epigenetic mutations have been discovered in GBM. It is critical to find and classify mutations in order to understand tumor behavior and therapy resistance throughout the clinical course. Glioblastoma multiforme is categorized into primary tumors developing from neural stem cell progenitors and secondary tumors arising from mutations in mature brain cells such as astrocytes due to the presence of various triggering mutations in addition to critical alterations in GBM stem cells.
Alterations in genetic information, which cause gene expression and suppression relative to their physiological levels in healthy brain cells, cause changes in both the cellular and extracellular matrix, resulting in a variety of biochemical patterns. Because of the amount of genotypic diversity, the word multiforme was established.
Glioblastoma symptoms and signs
In individuals with glioblastoma multiforme, it is critical to obtain a detailed history of symptoms. Symptoms vary depending on the location and size of the tumor and are comparable to those caused by benign or malignant brain tumors. GBM often manifests as a series of progressive neurological symptoms that last a few days to a few weeks. Seizures, focal neurological impairments, and symptoms linked with elevated intracranial pressure such as nausea, vomiting, impaired vision, and altered mental status are the most common symptoms. Physicians should have a high suspicion of malignancy due to vague symptoms that can also arise in infectious, inflammatory, or other disease processes.
Stage 4 Glioblastoma
Glioblastoma is not classified into stages. Brain malignancies are "graded" rather than "staged," with grades indicating how the cancer cells appear under a microscope as well as their tendency to reproduce.
Brain cancer grades, like stages, range from 1 to 4. The more aggressive the malignancy, the higher the grade. Glioblastomas, on the other hand, are invariably categorized as grade 4 brain cancer. This is due to the fact that this malignancy is a particularly aggressive variety of astrocytoma. Glioblastoma tumors are not staged because they all exhibit characteristics consistent with stage 4 brain cancer, such as:
- When viewed beneath a microscope, the cancerous cells look highly abnormal and have very few features of healthy cells.
- The cancerous cells reproduce at a rapid rate and often invade nearby parts of the brain.
- The tumors produce their own blood vessels to support the rapid growth, and the lesions may also contain calcium deposits and cystic material.
How Glioblastoma can be diagnosed?
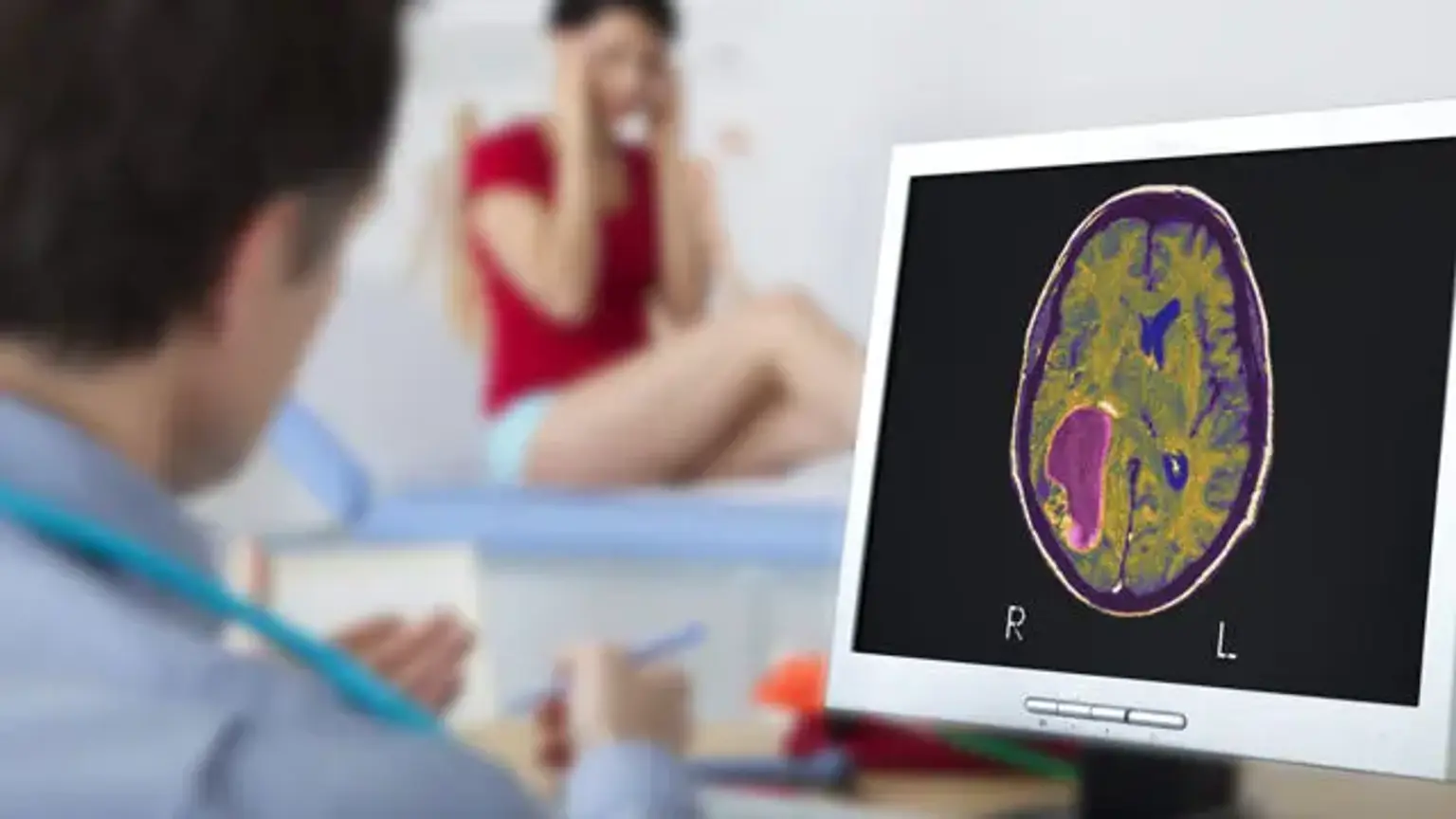
Glioblastomas are frequently seen as ring-enhancing lesions on MRI. However, the look is not specific, as different lesions such as abscess, metastasis, tumefactive multiple sclerosis, and other entities may seem similarly. A stereotactic biopsy or a craniotomy with tumor resection and pathologic confirmation is required for a definitive diagnosis of a suspected GBM on CT or MRI.
Because the tumor grade is determined by the most malignant region of the tumor, a biopsy or subtotal tumor resection may result in the lesion being undergraded. Perfusion MRI imaging of tumor blood flow and MR spectroscopy measuring tumor metabolite concentration may add diagnostic value to standard MRI in select cases by showing increased relative cerebral blood volume and increased choline peak, respectively, but pathology remains the gold standard for diagnosis and molecular characterization.
It is critical to distinguish primary glioblastoma from secondary glioblastoma. These tumors either develop spontaneously (de novo) or have progressed from a lower-grade glioma. Primary glioblastomas have a poor prognosis, diverse tumor biology, and may respond differently to therapy, making this an important evaluation to evaluate patient prognosis and therapy.
A mutation in IDH1 is found in more than 80% of secondary glioblastomas, although this mutation is seen in only 5–10% of initial glioblastomas. As a result, IDH1 mutations are a useful tool for distinguishing primary and secondary glioblastomas, because histopathologically they are quite similar and distinguishing them without molecular biomarkers is questionable.
Glioblastoma treatment
Treating glioblastoma is difficult due to several complicating factors:
- The tumor cells are resistant to conventional therapies.
- The brain is susceptible to damage from conventional therapy.
- The brain has a limited capacity to repair itself.
- Many drugs cannot cross the blood–brain barrier to act on the tumor.
Treatment of primary brain tumors consists of palliative (symptomatic) care and therapies intended to improve survival.
1. Symptomatic therapy:
Supportive treatment focuses on relieving symptoms and improving the patient's neurologic function. The primary supportive agents are anticonvulsants and corticosteroids.
Historically, around 90% of glioblastoma patients received anticonvulsant medication, despite the fact that only an estimated 40% of individuals required this treatment. Anticonvulsants should not be used prophylactically, according to neurosurgeons, who should wait until a seizure occurs before administering this medicine. Those who receive phenytoin alongside radiation may experience significant skin responses such as erythema multiforme and Stevens–Johnson syndrome.
Corticosteroids, typically dexamethasone, can reduce peritumoral edema (through reorganization of the blood–brain barrier), as well as mass effect and intracranial pressure, with a reduction in headache or drowsiness.
2. Surgery:
Surgery is the first stage of treatment of glioblastoma. An average GBM tumor contains 1011 cells, which is on average reduced to 109 cells after surgery (a reduction of 99%). Benefits of surgery include resection for a pathological diagnosis, alleviation of symptoms related to mass effect, and potentially removing disease before secondary resistance to radiotherapy and chemotherapy occurs.
The more extensive the tumor excision, the better. In retrospective studies, removing 98% or more of the tumor was associated with a much longer healthy life than removing less than 98 percent of the tumor. If the surgery is guided by a fluorescent dye known as 5-aminolevulinic acid, the odds of a near-complete first excision of the tumor are improved.
Because GBM cells are broadly infiltrative throughout the brain upon diagnosis, most persons with GBM have recurrent tumors, either near the original site or in more distant sites throughout the brain, despite "complete excision" of all apparent tumors. Following surgery, various modalities, mainly radiation and chemotherapy, are utilized to inhibit and halt recurring disease.
3. Radiotherapy:
Following surgery, radiation becomes the primary mode of treatment for persons with glioblastoma. It is usually done in conjunction with the administration of temozolomide.
A significant clinical research conducted in the early 1970s found that among 303 GBM patients randomly assigned to radiation or nonradiation therapy, those who got radiation had a median survival that was more than double that of those who did not. Following clinical research has attempted to expand on the foundation of surgery followed by radiation. After surgery, radiation can reduce the tumor size to 107 cells on average. When compared to more accurate and focused three-dimensional conformal radiotherapy, whole-brain radiotherapy does not improve.
GBM tumors are widely recognized to possess hypoxic zones of tissue that are extremely resistant to radiation. As of 2016, various techniques to chemotherapeutic radiosensitizers had been attempted with limited effectiveness. As of 2010, recent research approaches included preclinical and clinical studies exploring the use of an oxygen diffusion-enhancing chemical like trans sodium crocetinate as radiosensitizers, and a clinical trial was underway as of 2015. Boron neutron capture therapy has been investigated as an alternate treatment for glioblastoma, although it is not widely used.
4. Chemotherapy:
The majority of research demonstrate that adding chemotherapy has little benefit. However, in a large clinical trial of 575 persons randomized to normal radiation versus radiation plus temozolomide chemotherapy, the group getting temozolomide survived a median of 14.6 months versus 12.1 months for the group receiving radiation alone. This treatment regimen is now the standard of care for most GBM patients who are not involved in a research trial. Temozolomide appears to function by sensitizing tumor cells to radiation, and it appears to be more effective in tumors with methylated MGMT promoters.
In high-grade gliomas, high doses of temozolomide have minimal toxicity, but the effects are comparable to regular doses. Antiangiogenic therapy, such as bevacizumab, alleviates symptoms but has no effect on overall survival in glioblastoma patients. As of 2019, the total benefit of anti-angiogenic therapy is unknown. Concurrent and adjuvant chemoradiotherapy provides the best overall survival in elderly adults with newly diagnosed glioblastoma who are reasonably fit, but is linked with a higher risk of haematological side events than radiotherapy alone.
5. Cannabinoids:
Cannabinoids (cannabidiol derivatives) are known to be effective in oncology (via capsules of tetrahydrocannabinol (THC) or the synthetic analogue nabilone), on the one hand to combat nausea and vomiting caused by chemotherapy, and on the other hand to stimulate appetite and reduce the sense of anguish or actual pain. In mouse models, they have been shown to suppress growth and angiogenesis in malignant gliomas.
The findings of a pilot trial on the use of THC in end-stage patients with recurrent glioblastoma were promising and warranted additional investigation. Cannabinoids have been discovered to be able to assault the neoplastic stem cells of glioblastoma in animal models, resulting in on the one hand encouraging differentiation into more mature, potentially more "treatable" cells and on the other hand inhibiting carcinogenesis.
6. Other procedures:
Alternating electric field therapy is an FDA-approved treatment for glioblastoma, both newly diagnosed and recurrent. In 2015, initial reports from a phase-three randomized clinical trial of alternating electric field therapy plus temozolomide in newly diagnosed glioblastoma revealed a three-month improvement in progression-free survival and a five-month improvement in overall survival when compared to temozolomide therapy alone, making it the first large trial in a decade to show a survival improvement in this setting.
Despite these findings, the efficacy of this technique is still debated among medical specialists. However, increasing understanding of the mechanistic basis by which alternating electric field therapy exerts anti-cancer effects, as well as results from ongoing Phase 3 clinical trials in extracranial cancers, may help facilitate increased clinical acceptance of alternating electric field therapy to treat glioblastoma in the future.
Prognosis of Glioblastoma
The most typical length of survival after diagnosis is 12 to 15 months, with only 3 to 7% of persons living for more than five years. Between 2012 and 2016, the five-year survival rate in the United States was 6.8 percent. Without therapy, the average survival time is three months. Complete cures are extremely unusual, but they have occurred. There is now one person in the United States who has had the disease for more than 20 years.
Age (> 60 years) is associated with a worse prognosis. The most common cause of death is extensive tumor infiltration, which causes cerebral edema and elevated intracranial pressure.
Complications of Glioblastoma
In addition to the difficulties outlined above from chemotherapy and radiotherapy, the disease process itself has consequences such as recurrence. Pseudoprogression is a subacute deterioration of MRI abnormalities that occurs within three months of the conclusion of chemoradiotherapy. It is a side effect of the medication.
To avoid premature therapy termination, it is critical to distinguish between pseudoprogression and actual disease advancement. The primary distinguishing aspect is that pseudoprogression is frequently asymptomatic. If pseudoprogression is suspected, treatment should be continued unless the patient becomes symptomatic or clinical characteristics worsen.
Conclusion
Glioblastoma multiforme (GBM) is the most prevalent primary brain tumor in adults, accounting for 45.2 % of all malignant primary brain and central nervous system (CNS) tumors. GBM is still an incurable disease with a 15-month median survival. Only 5.5 % of patients lived five years after being diagnosed. GBMs are classified into primary and secondary subtypes that develop through different genetic paths, affecting patients at different ages with varying outcomes.
Primary GBMs account for 80% of GBMs and occur in older patients with a mean age of 62 years, whereas secondary GBMs arise in younger individuals with a mean age of 45 years from lower-grade astrocytoma or oligodendroglioma. Secondary GBMs are typically found in the frontal lobe, show less necrosis, and have a better prognosis than main GBMs.
The standard of care for a GBM is surgery, followed by six and a half weeks of daily radiation and oral chemotherapy, followed by a six-month schedule of oral chemotherapy administered five days each month. To begin, the neurosurgeon will remove as much of the tumor as feasible before implanting medicated wafers directly into the brain. These wafers breakdown naturally over time, gradually releasing chemotherapy medicines into the tumor area.
The FDA approved temozolomide, another chemotherapy medication, in 2013 and it is routinely used to treat GBMs and other advanced brain malignancies. The medication is administered as a tablet and acts by reducing tumor growth. Radiation therapy may be used to eliminate more tumor cells and treat cancers in people who are unable to undergo surgery.
The World Health Organization classifies GBM as a grade IV malignancy that is aggressive, mitotically active, and necrotic. The prognosis for GBM is dismal.