Introduction
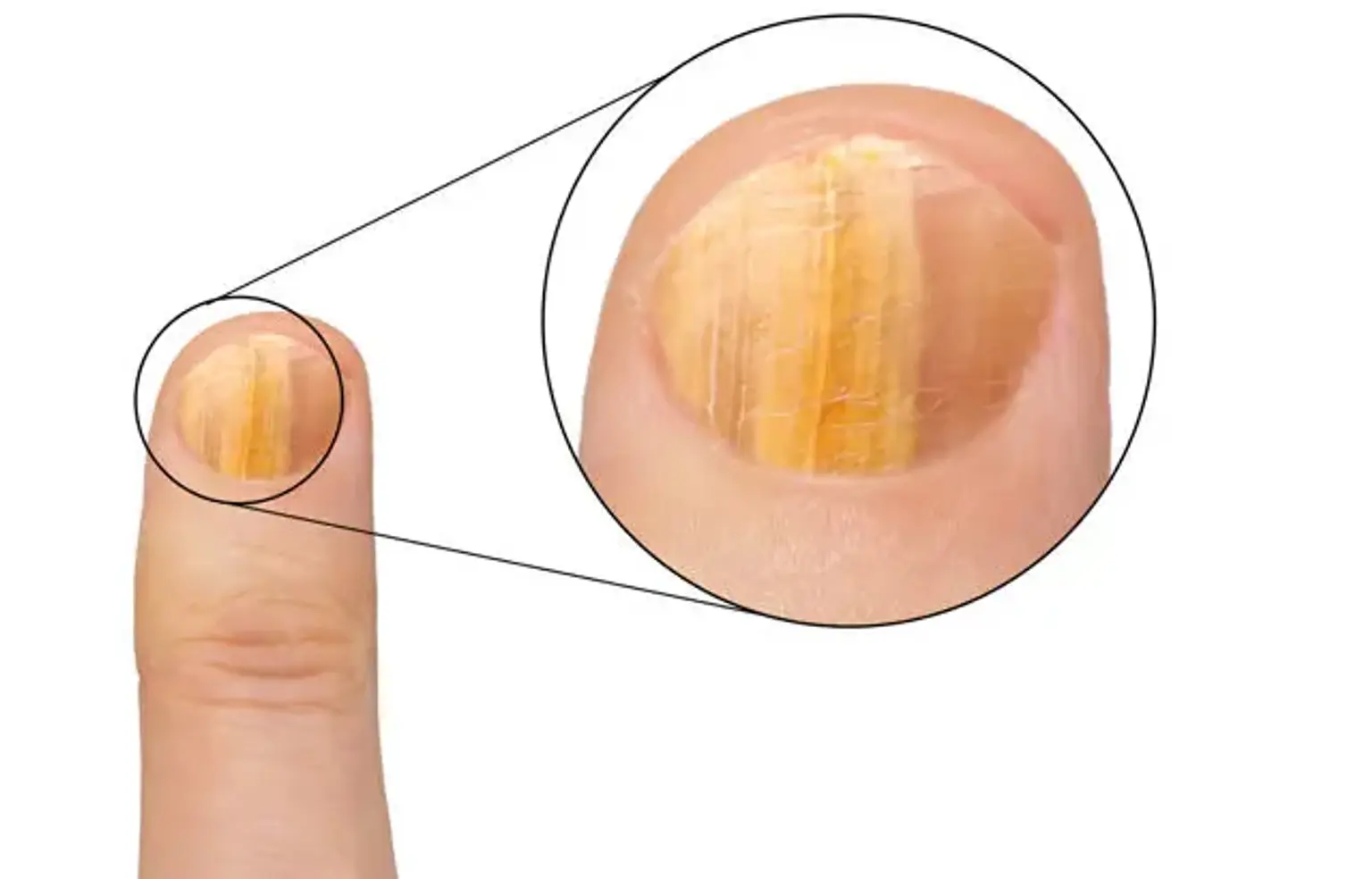
Nail fungus, medically known as onychomycosis, is a common fungal infection that affects the nails, causing them to become discolored, thickened, and brittle. This condition can occur in both toenails and fingernails but is more common in toenails due to the warm, moist environment inside shoes. Onychomycosis is often overlooked because it develops slowly, but if untreated, it can lead to permanent nail damage and may spread to other nails or even the skin.
The condition affects millions of people globally, with the risk increasing with age, poor circulation, and conditions such as diabetes. Early detection and treatment are crucial to avoid complications and improve the quality of life.
What is Onychomycosis?
Onychomycosis refers to a fungal infection that attacks the nail, specifically the nail bed and surrounding tissue. Fungal infections in the nails are often caused by dermatophytes (a type of fungi that thrives in warm, damp environments), but they can also be caused by molds and yeasts.
The infection can begin as a small patch of discoloration or a white or yellow spot under the nail. Over time, it may spread and cause the nail to become thick, brittle, and deformed. In more severe cases, the nail can become painful and separate from the nail bed.
Though this condition primarily affects adults, it can also occur in children, especially if they have compromised immune systems or a history of athlete's foot.
Causes of Nail Fungus (Onychomycosis)
Several factors can increase the likelihood of developing nail fungus. The primary cause is the overgrowth of fungi, typically dermatophytes, under or inside the nail. The infection occurs when the fungi enter the nail through small cuts, cracks, or other traumas.
Risk factors for developing onychomycosis include:
Age: Older adults are more prone due to slower nail growth and weakened immune systems.
Poor circulation: Conditions like diabetes or peripheral artery disease can reduce blood flow to the extremities, increasing the risk.
Injury to the nail: Damage to the nail or surrounding skin can provide an entry point for fungi.
Warm, moist environments: Fungi thrive in damp conditions, so wearing tight shoes, walking barefoot in public pools or gyms, and having sweaty feet increases susceptibility.
Weak immune system: Individuals with compromised immune systems, such as those with HIV or undergoing chemotherapy, are at higher risk.
Diagnosis of Nail Fungus
Diagnosing nail fungus typically begins with a physical examination by a healthcare provider. They may notice signs such as discoloration, thickening, or crumbling of the nail. However, to confirm the diagnosis and rule out other conditions, further tests may be necessary.
Common diagnostic methods include:
KOH test: A sample of the infected nail is scraped off and examined under a microscope after being treated with potassium hydroxide (KOH). This helps to identify the presence of fungal cells.
Fungal culture: A sample of the nail or skin is cultured in a lab to grow the fungi and identify the exact species causing the infection.
Biopsy: In rare cases, a small piece of the nail may be removed and examined in a lab to ensure the diagnosis is accurate.
Prompt diagnosis is essential for determining the appropriate treatment and preventing the infection from worsening.
Symptoms of Nail Fungus
Nail fungus, or onychomycosis, often starts with subtle symptoms that gradually worsen. The most common signs include:
Discoloration: The nail may turn white, yellow, or brown.
Thickening: The affected nail becomes thicker than normal and may feel uncomfortable or painful.
Brittleness and Crumbling: The nail becomes fragile, and pieces may break off or crumble easily.
Distortion: The nail may become distorted or misshapen, often growing out of alignment.
Foul Odor: An unpleasant smell can occur when the fungus spreads beneath the nail.
If left untreated, the infection can cause significant nail damage and may even spread to other nails or parts of the body. If you notice any of these symptoms, seeking treatment early can prevent further damage and improve outcomes.
Treatment Overview: Types of Nail Fungus Treatments
There are several options for treating nail fungus, each suited to different cases. The main categories include:
Oral Medications: These are typically used for more severe or widespread infections. Common oral antifungals like terbinafine and itraconazole work by targeting the fungus systemically, stopping it from growing and spreading.
Topical Treatments: These include antifungal creams, ointments, or lacquers that are applied directly to the infected nail. While effective for mild cases, they often take longer to show results than oral medications.
Laser Treatment: A newer treatment option, lasers target the fungus with focused light, killing the fungi beneath the nail without affecting surrounding tissue.
Each treatment method has its pros and cons, and the best choice depends on the severity of the infection, the type of fungus involved, and individual health factors.
Oral Antifungal Medications
Oral antifungal medications are often the first line of treatment for moderate to severe onychomycosis. These drugs work by attacking the fungal infection from within the body. Popular oral antifungals include:
Terbinafine (Lamisil): One of the most commonly prescribed oral antifungals, terbinafine works by stopping the fungi from producing ergosterol, an essential component of the fungal cell membrane.
Itraconazole (Sporanox): This medication inhibits fungal growth by affecting the cell membrane, similar to terbinafine.
Fluconazole (Diflucan): Another antifungal, often used when other medications are not effective.
Treatment duration typically lasts 6 to 12 weeks, depending on the medication and the severity of the infection. While these medications are effective, they may come with side effects, such as liver damage, gastrointestinal issues, or headaches, so regular monitoring by a doctor is necessary.
Topical Antifungal Treatments
Topical treatments are often preferred for mild to moderate cases of nail fungus. These products, which come in the form of creams, gels, or nail lacquers, are applied directly to the affected nail. Some common topical antifungal options include:
Ciclopirox (Penlac): A nail lacquer that’s applied daily to the infected nail. It’s effective in treating superficial nail infections.
Efinaconazole (Jublia): A newer topical treatment that is applied directly to the nail surface once daily.
Amorolfine: A topical lacquer commonly used in Europe, it works by disrupting the fungal cell membrane.
While effective for many, topical treatments require persistence, as they can take several months to show visible results. The fungus needs to be completely eradicated from the nail, which means continuous application and patience.
Laser Treatment for Nail Fungus
Laser therapy has emerged as a promising treatment option for onychomycosis. Unlike oral or topical medications, laser treatment uses focused light to target and kill the fungi beneath the nail without harming surrounding tissue. The most common laser used for nail fungus is the Nd:YAG laser, which penetrates the nail and heats the fungal cells, causing them to break down.
Laser treatment is often considered for cases that are resistant to oral or topical medications, or when patients are unable to take oral antifungals due to health concerns. It’s generally quick, with each session lasting about 30 minutes, and typically requires a series of treatments spaced a few weeks apart.
While studies show promising results, laser therapy is still considered a supplementary treatment, and success rates may vary depending on the severity of the infection.
Home Remedies for Nail Fungus
Before seeking professional treatment, some individuals try home remedies to manage mild nail fungus. Although these methods are not scientifically proven to be as effective as medical treatments, some may help reduce symptoms or slow the infection:
Tea Tree Oil: Known for its antifungal properties, tea tree oil can be applied directly to the infected nail. Some studies suggest it may help with fungal infections, although its effectiveness is limited for more severe cases.
Vinegar Soaks: A diluted vinegar soak (mixing one part vinegar to two parts water) is a popular home remedy thought to kill the fungus. However, it is not as effective as antifungal medications.
Garlic: Garlic contains allicin, which has antifungal properties. Some people apply crushed garlic directly to the affected nail or add it to their diet.
While these remedies may provide some relief, they are not substitutes for professional medical treatments, especially for more persistent or severe cases.
When to Seek Professional Help for Nail Fungus
It can be tempting to try home remedies or ignore the problem, especially if the symptoms are mild. However, it’s important to seek professional medical attention if:
Symptoms persist or worsen despite using over-the-counter antifungals or home treatments.
Pain or discomfort occurs, especially if the nail becomes swollen or infected.
Nail damage becomes severe, making it difficult to wear shoes or perform daily tasks.
The infection spreads to other nails or to the skin.
A healthcare provider can recommend the most effective treatment based on the severity of the infection and your medical history. Early treatment can prevent the infection from spreading and reduce the risk of permanent nail damage.
Preventing Nail Fungus: Tips and Best Practices
Prevention is key to avoiding nail fungus, especially for those who are prone to it. Here are some practical tips to reduce the risk:
Keep your nails dry and clean: Fungi thrive in moist environments, so ensure your feet and nails are dry after washing. Dry your nails thoroughly after showers or swimming.
Wear breathable footwear: Avoid wearing tight, non-breathable shoes for long periods. Opt for shoes that allow airflow to your feet, especially in warm climates.
Use antifungal powders or sprays: If you're in environments like gyms or public swimming pools, applying antifungal powder to your feet can help prevent infections.
Avoid sharing personal items: Never share nail clippers, towels, or shoes with others, as this can spread fungal infections.
Trim nails regularly: Keep nails short and trimmed straight across to avoid trapping moisture or dirt underneath.
Good hygiene, combined with these preventive measures, can significantly reduce the risk of developing nail fungus.
Risks and Complications of Untreated Nail Fungus
If left untreated, nail fungus can lead to several complications, affecting not just the appearance but also the health of the nail and surrounding skin. Some potential risks include:
Permanent Nail Damage: Over time, the infection can cause the nail to become thickened, brittle, and deformed. In severe cases, the nail may completely detach from the nail bed, which can be painful and difficult to treat.
Spread to Other Nails: Onychomycosis can spread from one nail to others, especially if the infection isn’t managed properly. It can also spread to the skin, leading to athlete’s foot or fungal infections in other parts of the body.
Increased Risk of Secondary Infections: The damaged nail and surrounding tissue are more vulnerable to bacterial infections, which can cause further complications, including cellulitis (a skin infection) or abscesses.
Chronic Recurrence: Without treatment, the infection can become chronic, with recurring flare-ups that are harder to treat and may require stronger or longer courses of medication.
Because of these potential complications, early intervention is important for both cosmetic and health reasons.
Nail Fungus Treatment: What to Expect During Recovery
Recovery from nail fungus treatment varies depending on the treatment method, the severity of the infection, and how early the infection was addressed. Here's what to expect during the recovery process:
Oral Medications: For those using oral antifungal medications, recovery typically takes several weeks to months. Patients may begin to notice improvements in the appearance of their nails within the first few weeks, but full recovery can take 6 to 12 months, as nails grow slowly.
Topical Treatments: These may take longer to show results, sometimes up to 12 months, as the nail must fully regrow to clear the infection completely. The affected area should be regularly cleaned and treated as per the instructions.
Laser Treatment: Patients may experience faster recovery, with visible improvements seen after a few sessions. However, it may take several months for the new nail to fully grow out and look normal.
During recovery, it’s important to follow all treatment guidelines and take preventative measures to avoid re-infection.
Costs of Nail Fungus Treatment
The cost of treating nail fungus can vary widely depending on the treatment method, the severity of the infection, and the region in which you live. Below is a general breakdown of potential costs:
Oral Medications: Prescription oral antifungals typically cost between $30 to $200 per month, depending on the brand and dosage. For a complete 3-month course, you may pay anywhere from $90 to $600.
Topical Treatments: Over-the-counter topical antifungal treatments can range from $10 to $50 for a month’s supply. Prescription treatments like ciclopirox nail lacquer may cost $100 or more for a monthly supply.
Laser Treatment: Laser therapy can be expensive, ranging from $500 to $1,000 per session, with multiple sessions required for effective treatment. The total cost for laser treatment could be anywhere from $1,500 to $4,000 or more, depending on the clinic.
In some cases, insurance may cover part of the costs, particularly for more severe infections. Always check with your provider for details about coverage.
Myths and Misconceptions About Nail Fungus
There are several myths surrounding nail fungus, which can lead to confusion and improper treatment. Let’s debunk some common misconceptions:
Myth 1: Nail fungus is only a cosmetic problem. While it may start as a cosmetic issue, untreated nail fungus can cause permanent nail damage, pain, and secondary infections. It’s more than just an aesthetic concern.
Myth 2: Nail fungus will go away on its own. Fungal infections do not resolve on their own and typically worsen over time without treatment. Early intervention is crucial to prevent the infection from spreading or becoming chronic.
Myth 3: Home remedies are enough to cure nail fungus. While some home remedies, like tea tree oil, may offer mild relief, they are not proven to cure nail fungus completely. Professional treatments are necessary for effective and lasting results.
Myth 4: Only people with poor hygiene get nail fungus. Nail fungus can affect anyone, regardless of hygiene. People with compromised immune systems, poor circulation, or those frequently exposed to damp environments are at higher risk.
Understanding the facts and dispelling myths about nail fungus can help people make informed decisions about their treatment options.
Nail Fungus and Diabetes: Special Considerations
People with diabetes are at an increased risk of developing nail fungus due to poor circulation and a weakened immune system. Elevated blood sugar levels also create an ideal environment for fungal growth. Diabetic individuals should be especially cautious about foot health and nail care to prevent complications.
Untreated nail fungus can lead to more serious issues for diabetics, such as infections that may not heal well or spread quickly. If you have diabetes and notice signs of nail fungus, it's essential to seek treatment promptly. Regular monitoring and good foot hygiene can help prevent fungal infections from worsening.
Lifestyle Changes to Support Nail Health
Maintaining healthy nails goes beyond treatment – lifestyle changes can help prevent fungal infections. Some tips include:
Foot hygiene: Wash feet daily with soap and water, and dry thoroughly, especially between the toes.
Nail care: Trim nails regularly, avoid cutting them too short, and use clean tools to avoid introducing fungi.
Breathable footwear: Opt for shoes made of natural materials like leather or mesh that allow the feet to breathe.
Avoid damp environments: If possible, avoid walking barefoot in public places like swimming pools or gyms, where fungi thrive.
These practices reduce the risk of fungal infections and support overall nail health.
Prognosis and Long-Term Management of Nail Fungus
With proper treatment, nail fungus can be effectively managed, though full recovery may take several months due to the slow growth of nails. Some people may experience recurrent infections, especially if they do not follow preventive measures or complete their prescribed treatment regimen.
In many cases, once the infection clears, nails grow back healthy, but it’s crucial to maintain good hygiene to prevent reinfection. If the fungus recurs, alternative treatments or more aggressive therapies like laser treatment may be needed.
Conclusion
Nail fungus, while common, should not be ignored. Early diagnosis and treatment are key to avoiding complications and restoring the health of your nails. Whether through oral medications, topical treatments, or laser therapy, there are effective options to treat onychomycosis. By understanding the causes, risks, and treatment options, you can make informed decisions and improve your nail health.
If you suspect you have nail fungus, seek professional advice to determine the best course of action. With proper care and treatment, you can prevent the infection from worsening and regain healthy, beautiful nails.