Pancreaticoduodenectomy
Overview
Despite advances in medical treatment, chemotherapy, radiation, and molecular biology, pancreatic cancer is the fourth leading cause of cancer mortality, with a 5-year survival rate of just approximately 12%. The pancreatoduodenectomy, often known as the Whipple surgery, is the surgical method of choice for pancreatic ductal adenocarcinomas that are resectable or borderline resectable.
Because organs in the proximal gastrointestinal system share a blood supply, surgical removal of the head of the pancreas entails removal of the duodenum, proximal jejunum, gallbladder, and, in certain cases, a portion of the stomach.
Pancreaticoduodenectomy definition
The most common operation for treating pancreatic cancer is a pancreaticoduodenectomy, often known as the Whipple procedure. The treatment is named for Allen Oldfather Whipple, former chairman of the Department of Surgery at Columbia University and the method's inventor. It is used to treat tumors at the head of the pancreas, which accounts for around 75% of pancreatic cancer tumors.
Dr. Alessandro Codivilla, an Italian surgeon, conducted the first pancreaticoduodenectomy in 1898, which was later improved by Dr. Walter Kausch in 1912. The operation was then conducted in two stages, then Dr. Allen Whipple modified it in 1940 to become a one-stage Whipple technique.
Later, it was discovered that this operation is associated with a high death rate; however, with improved medical understanding and surgical innovation, mortality has decreased significantly. In high-load hospitals, the mortality rate lowers to roughly 3% to 5% when a skilled surgeon is present.
Pancreatoduodenectomies are classified into two types: the more extensive "classical Whipple (CW)" and the "pylorus sparing pancreatoduodenectomy (PSD)." The pancreatic head, the duodenum, a segment of the stomach, the gallbladder, and a portion of the bile duct are all removed in the CW, but the portion of the stomach is spared in the PSD.
Furthermore, pancreaticoduodenectomy can be performed laparoscopically as well as openly. Despite this, new studies on the feasibility and shorter postoperative results of both open and laparoscopic pancreatoduodenectomy have revealed that the laparoscopic method is associated with shorter length of stay, less blood loss, and better lymph node dissection.
However, clinicians all over the world are still hesitant to use the laparoscopic method, which is most likely due to the exceedingly difficult dissection and anastomosis in this surgery. Furthermore, the oncological outcomes of the open and laparoscopic pancreatoduodenectomy strategies are still hazy, if not doubtful.
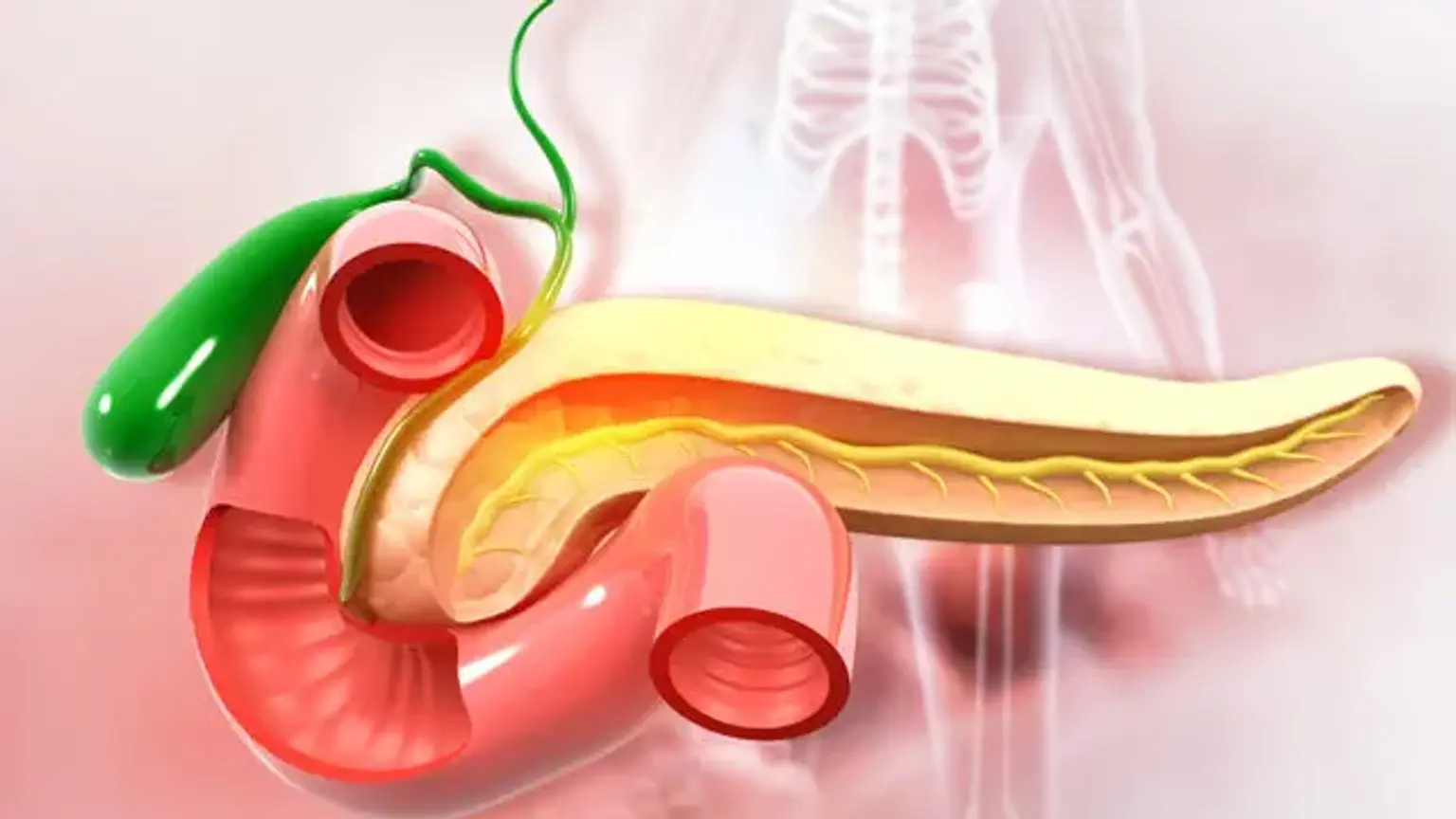
Anatomy involved in the procedure
The most frequent pancreaticoduodenectomy approach involves removing the distal segment (antrum) of the stomach, the first and second segments of the duodenum, the head of the pancreas, the common bile duct, and the gallbladder all at once. During the procedure, the lymph nodes in the region are frequently removed as well (lymphadenectomy).
However, in the most frequent kind of pancreaticoduodenectomy, not all lymph nodes are removed since studies demonstrate that patients do not benefit from the more thorough operation.
The surfaces of the peritoneum and the liver are evaluated for metastasized illness at the very beginning of the surgery, when the surgeons have acquired access to the abdomen. This is a critical initial step since the presence of active metastatic illness precludes the procedure from being performed.
The pancreas receives circulatory supplies from the celiac artery via the superior pancreaticoduodenal artery and from the inferior pancreaticoduodenal artery via the superior mesenteric artery. The right gastric artery, which is similarly derived from the celiac artery, produces additional smaller branches. The excision of the duodenum and pancreatic head is necessary since they share the same arterial blood supply (the superior pancreaticoduodenal artery and inferior pancreaticoduodenal artery).
Because these arteries flow via the head of the pancreas, if the single blood supply is destroyed, both organs must be removed. If only the head of the pancreas was removed, blood supply to the duodenum would be compromised, resulting in tissue necrosis.
The common bile duct is removed while the blood supply to the liver is preserved. This implies that, as long as the liver has a healthy blood supply, the surgeon will need to create a new link to drain the bile generated by the liver. This is done at the conclusion of the procedure. A new connection will be formed between the pancreatic duct and the jejunum or stomach by the surgeon. A cholecystectomy is performed during the operation to remove the gallbladder. The gallbladder is removed individually, therefore this section is not performed as a whole.
The important arterial structures in the region that were not removed during the treatment include the portal vein, the superior mesenteric vein, and the superior mesenteric artery, as well as the inferior vena cava. These structures are critical to consider in this procedure, especially if it is being performed to remove a tumor from the head of the pancreas.
If the tumor encases (wraps around 50% or more of the vessel) the celiac artery, superior mesenteric artery, or inferior vena cava, the procedure is termed unresectable owing to the lack of patient benefit while carrying a significant risk. A segment of the superior mesenteric vein or portal vein is occasionally connected to or inseparable from the tumor. Vascular surgeons resect the affected segment of the artery in this circumstance, and the vessel is repaired either by end-to-end anastomosis, repair of the vein's side wall, or a vein graft.
Indications
Pancreaticoduodenectomy is most commonly used to treat periampullary cancer, which includes cancer of the bile duct, duodenum, or pancreatic head. The pancreas, duodenum, and common bile duct all share a blood supply, necessitating en bloc excision of these several tissues. Chronic pancreatitis, benign pancreatic tumors, cancer metastatic to the pancreas, multiple endocrine neoplasia type 1, and gastrointestinal stromal tumors are all reasons for a pancreaticoduodenectomy.
Pancreatic cancer
Pancreaticoduodenectomy is the only possibly curative operation for pancreatic cancer. However, because the majority of pancreatic cancer patients have metastatic or locally advanced unresectable disease, only 15-20% of patients are candidates for the Whipple treatment.
Surgery may be performed after neoadjuvant chemotherapy, which seeks to reduce the tumor and increase the chances of total resection. Post-operative death and complications following pancreaticoduodenectomy have grown less prevalent, with rates of post-operative mortality dropping from 10-30% in the 1980s to less than 5% in the 2000s.
Cholangiocarcinoma
When cholangiocarcinoma, or cancer of the bile duct, is found in the distal biliary system, mainly the common bile duct that empties into the duodenum, it is an indication for the Whipple surgery. Curative surgical resection of cholangiocarcinoma may entail hepatectomy, or removal of a portion of the liver, with or without pancreaticoduodenectomy, depending on the location and extent of the cholangiocarcinoma.
Chronic pancreatitis
Chronic pancreatitis is often treated with pain management and exocrine insufficiency treatment. The major surgical rationale for surgical therapy of chronic pancreatitis is intractable abdominal discomfort. The removal of the pancreas head can reduce pancreatic duct blockage caused by chronic pancreatitis.
Trauma
It is unusual for forceful abdominal trauma to cause pancreatic and duodenum damage. When this pattern of injuries has been observed, it has usually occurred as a result of a lap belt in a car collision. When abdominal trauma causes bleeding around the pancreas and duodenum, injury to the common bile duct, pancreatic leakage, or duodenum transection, a pancreaticoduodenectomy is done. Due to the rarity of this operation in the context of trauma, there is insufficient data to support post-operative results.
Contraindications
The operation is contraindicated in the presence of changeable variables. It is also crucial to grasp the resectability criteria in order to better appreciate the contraindications. For localized pancreatic ductal adenocarcinoma, the three resectability grades are resectable, borderline resectable, and unresectable.
Localized and Resectable
There was no distant metastases, no indication of portal vein (PV) or superior mesenteric vein (SMV) distortion on radiographs, and clean dissection planes around the celiac axis, hepatic artery, and superior mesenteric artery (SMA)
B- Borderline Resectable
SMV-PV involvement characterized by deformation, constriction, and occlusion, but the presence of appropriate proximal and distal arteries for repair. Encasement of the gastroduodenal artery up to the hepatic artery with a brief segment encasement or abutment of the hepatic artery without reaching to the celiac axis. Tumor abutment of the superior mesenteric artery that does not extend beyond 180 degrees of the arterial wall circumference.
C- Unresectable for Cancer of the Head (Contraindication of the Procedure)
- Distance metastasis
- Tumour encasement of SMA more than 180 degrees
- Celiac abutment
- IVC involvement
- Aortic involvement
- Irreparable SMV or PV occlusion
Technique
A vertical midline incision or a bilateral subcostal incision is used for pancreaticoduodenectomy (PD). A retractor with self-retention is used. The initial step in pancreaticoduodenectomy is a detailed assessment of disease extent to determine resectability. Staging laparoscopy is now commonly used to evaluate resectability, and it can be done as a standalone surgery or in conjunction with PD. The whole liver is examined and palpated to check for the existence of metastases.
Intraoperative ultrasonography might be used to assess any worrisome abnormalities on the liver that are ambiguous on palpation. The parietal and visceral peritoneal surfaces, the Treitz ligament, the omentum, and the whole small and large intestine are checked for metastases. Lymph node involvement in the celiac axis is examined. Any lymph nodes that appear worrisome outside of the targeted field of dissection are frozen sectioned.
If the biopsy reveals that the malignancy has spread, PD is not done. By mobilizing the gallbladder and dissecting the cystic duct down to the junction of the common hepatic and common bile ducts, the porta hepatis is examined. The existence of tumor tissue is checked in the appropriate hepatic artery and the common hepatic artery.
The Kocher technique involves raising the duodenum and pancreatic head out of the retroperitoneum. Except for mobilization and excision of SMV or if the tumor extends into the 4th segment of the duodenum, the Cattell-Braasch procedure (extensive mobilization of the whole right colon and small intestine mesentery) is typically not required.
The gallbladder is separated from the liver, and the distal common hepatic duct is split around the cystic duct entrance point. The proximal bile duct is left unclamped to minimize crush damage, whereas the distal bile duct is clamped or sutured to prevent bile and tumor cell spilling. The bile duct is withdrawn caudally, and portal dissection is continued at the portal vein's anterior side.
During these techniques, the portal structures should be evaluated and safeguarded for a replacement right hepatic artery. If the patient looks to have an accessory right hepatic artery as well as a substantial native right hepatic artery, the auxiliary vascular can frequently be removed without causing any complications. To reduce the risk of erosion and bleeding, the gastroduodenal artery is ligated. The division of the gastroduodenal artery also aids in exposing the anterior side of the portal vein and facilitating portal vein dissection beyond the pancreatic neck.
The portal vein is located above the pancreas's neck, while the SMV is located below the pancreas's neck. The goal of blunt dissection downwards along the portal vein is to establish a plane in front of the portal vein and beneath the pancreas neck. This plane is critical in the operation, and if a clean plane cannot be created between the portal vein and the pancreatic neck, the procedure should be aborted.
Extensive Kocher maneuvering should expose the SMV inferior to the pancreatic neck. As it takes tributaries from the uncinate process and neck of the pancreas, the larger curvature of the stomach, and the transverse mesocolon, the superior mesenteric vein travels anterior to the third portion of the duodenum and is bordered by adipose tissue.
Following the middle colic veins to their juncture with the superior mesenteric vein or tracing the course of the right gastroepiploic vein to the point where it meets the superior mesenteric vein immediately below the inferior border of the pancreas are two ways to identify the superior mesenteric vein. Under direct eyesight, the plane anterior to the superior mesenteric vein is produced, but the splenic vein is retained. The intersection of the PV and SMV should be readily visible.
If required, a vascular resection is done. Typically, vascular surgeons perform segmental excision of the portal vein or superior mesenteric vein. Primary repair, patch closure, or an autologous graft (internal jugular vein, left renal vein, splenic vein, or larger saphenous vein) or a synthetic graft can be used to seal the vascular lesion (PTFE). Postoperatively, the repaired portal vein should be monitored for the possibility of portal vein thrombosis.
Antrectomy is performed for the Whipple operation by splitting the right gastric and right gastroepiploic arteries and dividing the antrum using a linear stapler. A pylorus-preserving pancreaticoduodenectomy is performed in which the proximal GI tract is split 2 to 3 cm distal to the pylorus with a linear stapling device.
The jejunum is split 15 to 20 cm distal to the Treitz ligament and brought in dorsal to the superior mesenteric arteries from left to right. A Penrose drain is looped beneath the pancreatic neck in the constructed tunnel to elevate the pancreas neck and protect the underlying portal vein.
After ensuring a free plane anterior to the portal and superior mesenteric veins, the pancreatic neck is separated using electrocautery. Stay sutures are inserted superiorly and inferiorly on the pancreatic remnant to prevent bleeding from the segmental pancreatic arteries that pass through those areas.
The specimen is solely linked by the pancreatic uncinate process. It is distinguished from the portal vein, superior mesenteric vein, and superior mesenteric artery by its location. This is accomplished by clamping, splitting, and tying the smaller branches of the portal and superior mesenteric vessels in a serial fashion.
The specimen is taken in one piece, with the pancreatic neck margin, uncinate margin, and common hepatic duct edges labeled for pathologists. The common hepatic duct margin and the pancreatic neck margin are critical and should be frozen sectioned early to evaluate the amount of future resection.
Following the excision of the specimen, metal clips are put at the resection field's margins to focus postoperative radiation therapy. Reconstruction following pancreaticoduodenectomy can be accomplished using a variety of techniques. To create GI continuity, the pancreatic anastomosis (PA) is performed first, followed by the bile duct and, ultimately, the duodenum.
PA is the most important step in pancreaticoduodenectomy, and it is performed with great care to reduce morbidity. For the rebuilding of the pancreatic anastomosis, either the jejunum or the stomach might be employed. PA can be built in two ways: duct to mucosa anastomosis or invagination method. The duct to mucosa anastomosis is built in a retrocolic way between the end of the pancreatic duct remnant and the side of the jejunum for the pancreaticojejunostomy.
The pancreatic duct is junked into the jejunum for the invagination method. Pancreaticojejunostomy procedures include modified Blumgart, modified Kakita, binding pancreaticojejunostomy anastomosis, and so on. The diversity of various procedures helps to enhance the pace of pancreatic fistula development, but no single way is optimal.
Following the repair, an end-to-side hepaticojejunostomy is done 10 to 15 cm distal to the pancreaticojejunostomy using interrupted single layer synthetic absorbable suture. The gastrojejunostomy is the third anastomosis done in patients who have had conventional pancreaticoduodenectomy with distal gastrectomy or duodenojejunostomy in situations of pylorus preservation. This anastomosis is usually done 10 to 15 cm downstream from the hepaticojejunostomy, proximal to the jejunum across the mesocolon defect.
Closed suction drains are placed at the Morrison pouch and near the pancreatic and biliary anastomosis after the reconstruction is completed. A feeding jejunostomy is frequently unnecessary and is at the discretion of the surgeon. The postoperative care comprises giving the patient nothing by mouth for the first day and then gradually transitioning to a low-fat diet in frequent tiny meals.
A nasogastric tube is used to decompress the stomach overnight following surgery, and it is generally withdrawn the next morning if there is no excessive output. Once the output is limited, there is no sign of pancreatic fistula, and the patient can tolerate a regular diet, the drains are progressively withdrawn.
Complications
- Delayed gastric emptying is described as the inability to remove the nasogastric tube and accept a solid food following surgery.
- Pancreatic fistula, defined as drain output with an amylase concentration greater than three times the upper limit of serum amylase on postoperative day 3 or later.
- Postpancreatectomy hemorrhage
- Wound infection
- Intra-abdominal abscess
Clinical Significance
Despite advances in medical treatment, chemotherapy, radiation, and molecular biology, pancreatic cancer is the fourth leading cause of cancer mortality, with a 5-year survival rate of just approximately 12%. The only curative therapy is pancreaticoduodenectomy. The late detection of cancer due to a lack of distinct symptoms makes this malignancy one of the most fatal. Finding diagnostic markers to detect pancreatic cancer in its early stages is critical.
Pancreaticoduodenectomy and its consequences have a significant morbidity and death rate. Because of the substantial morbidity associated with this operation, more careful postoperative care is required. A high level of suspicion and early diagnosis of postoperative complications is crucial in the patient's treatment. Physicians, nurses, and the whole team should be properly educated, and an organized team approach to patient care should be applied.
Conclusion
The Whipple procedure, also known as pancreaticoduodenectomy, is the most common surgery used to remove pancreatic tumors. Surgery to remove a tumor provides the best chance of long-term management of all kinds of pancreatic cancer. The Whipple is a demanding and complex procedure that removes and reconstructs a considerable portion of the gastrointestinal system.
The most frequent problem following a Whipple surgery is delayed stomach emptying. This signifies that the stomach takes an abnormally long time to expel its contents. After 7-10 days, the stomach usually begins to empty sufficiently to allow recovery. If the patient's delayed stomach emptying persists, he or she may require feedings through a feeding tube or vein for several weeks.
The most significant possible consequence of a Whipple surgery is an abdominal infection caused by pancreatic leaking into the small intestine. This is known as a pancreatic leak or a pancreatic fistula. This happens in around 10% of patients and is typically treated with a mix of drainage tubes and antibiotics. Many of these patients are in need of dietary assistance.
Before your Whipple procedure, your surgeon will discuss what to anticipate before, during, and after surgery, as well as any potential dangers. Your treatment team will discuss how your operation will influence your quality of life with you and your family. Chemotherapy, radiation treatment, or both may be used before or after the Whipple surgery or other pancreatic procedures for cancer. Discuss any concerns you have about your surgery and alternative treatment choices with your doctor, either before or after the procedure.
Before being admitted to the hospital, discuss your hospital stay with your family or friends and any assistance you may require from them when you return home. You will want assistance during the first few weeks following being discharged from the hospital. When you return home, your doctor and treatment team may give you guidelines to follow during your recuperation.