Parathyroid Disease
Overview of Parathyroid Disease
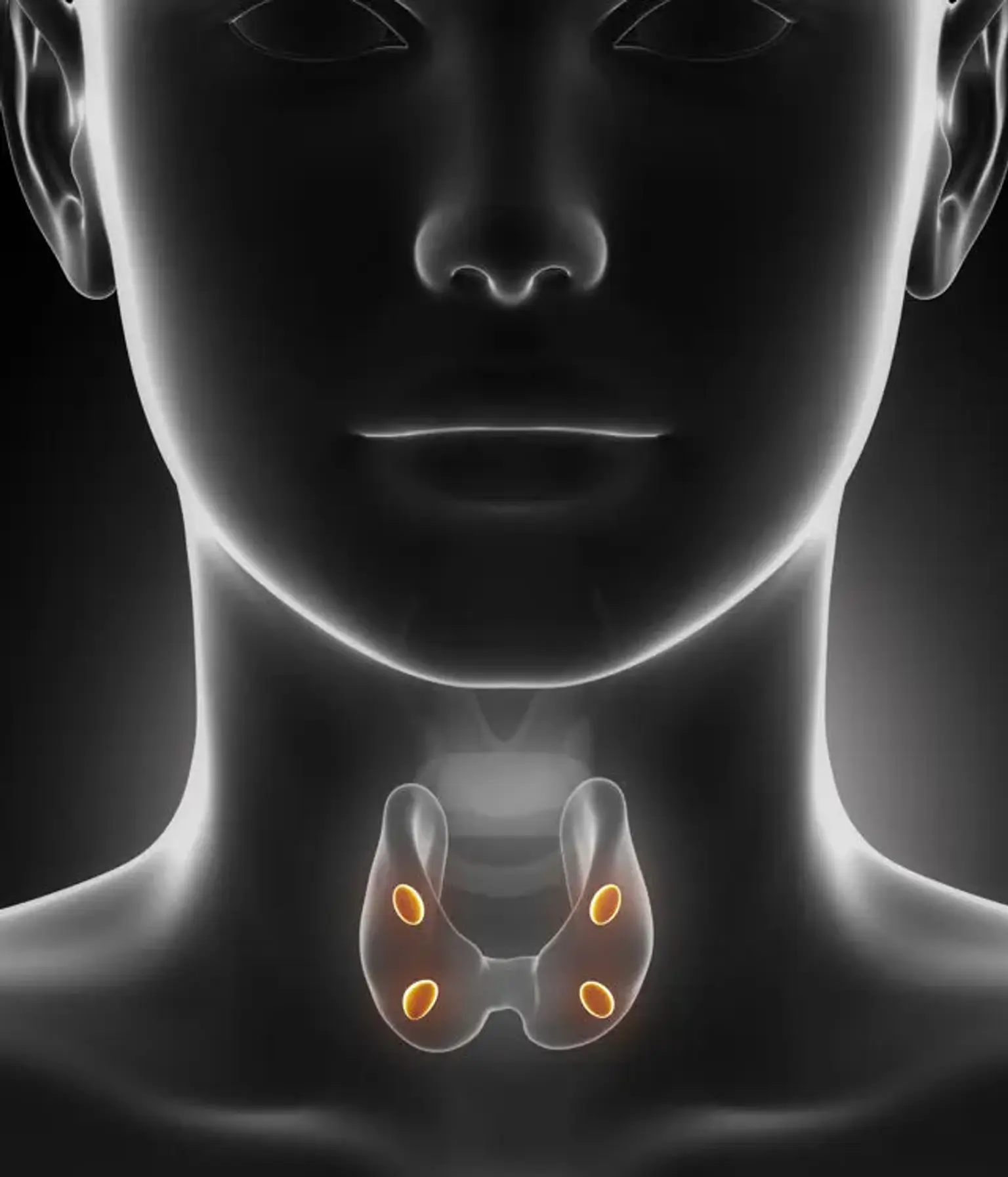
The parathyroid glands are located in the neck, right beneath the thyroid glands. The parathyroid glands (light pink) secrete parathyroid hormone, which raises calcium levels in the blood.
The most frequent symptom of a parathyroid gland disorder is an increase in blood calcium levels. Primary hyperparathyroidism, the most prevalent cause of hypercalcemia in outpatients, is generally asymptomatic or may be associated with bone disease, nephrolithiasis, or neuromuscular symptoms. Chronic kidney disease patients may develop secondary hyperparathyroidism, resulting in chronic kidney disease-mineral and bone conditions.
Hypoparathyroidism is most commonly caused by neck surgery; however, it can also be caused by autoimmune destruction of the glands and other less prevalent issues. A history and physical examination are performed on individuals with abnormal blood calcium levels, as is a repeat test of serum calcium levels, as well as measurements of creatinine, magnesium, vitamin D, and parathyroid hormone levels.
Parathyroidectomy is the therapy for symptomatic primary hyperparathyroidism. Asymptomatic primary hyperparathyroidism is managed by closely monitoring symptoms, blood calcium and creatinine levels, and bone mineral density. Hypoparathyroidism needs constant monitoring and vitamin D (calcitriol) therapy.
There are three types of parathyroid disease: hyperparathyroidism, parathyroid cancer, and hypoparathyroidism.
Parathyroid Adenoma
In the mid-1880s, the parathyroid glands were discovered in mammals. In 1925, Felix Mandl conducted the first parathyroidectomy in Vienna. There have been numerous breakthroughs in the knowledge, diagnosis, and treatment of parathyroid illness since then.
The parathyroid glands are tiny oval-shaped structures located near the thyroid. Eighty-five percent of individuals have four glands, two superior and two inferior. Superior glands (derived from the fourth branchial pouch) are often found on the posterior-lateral surface of the middle to superior thyroid lobe.
Because of their embryological descent with the thymus (both originating from the third branchial pouch), the inferior glands can be discovered at any point along their line of development. They are most typically found in the inferior portion of the thyroid gland.
A healthy parathyroid gland is around the size of an apple seed and weighs about 0.5 g. Microadenomas are tumors that weigh less than 0.1 g, whereas a big adenoma weighs more than 2 g. An adenoma typically weighs 1 g.
Etiology
A parathyroid adenoma is one of several types of parathyroid illness, including parathyroid hyperplasia and parathyroid cancer.
For the majority of individuals, the cause of a parathyroid adenoma is unknown. The cyclin D1/PRAD1 gene is the most commonly related genetic mutation with sporadic adenomas. PTH secretion is affected when this gene's normal function is disrupted. Approximately 20 to 40% of spontaneous adenomas contain cyclin D1 overexpression.
In terms of environmental variables, some evidence suggests that a patient's history of radiation therapy predisposes them to parathyroid illness later in life. Long-term calcium deprivation can potentially cause parathyroid illness owing to prolonged PTH activation.
Clinical presentation
Many people with a parathyroid adenoma are asymptomatic, with hypercalcemia detected by chance during normal blood testing. Symptoms of hypercalcemia, such as bone pain, tiredness, polyuria, nephrolithiasis, constipation, and neuropsychiatric disturbance, are the most prevalent manifestations of symptomatic parathyroid illness.
Calcium elevations that are too high might result in problems such as cardiac arrhythmias, unconsciousness, and death. A patient is unlikely to have visible symptoms of a parathyroid adenoma. The diseased gland is seldom palpable, and a visible neck lump suggests thyroid pathology or parathyroid malignancy.
Evaluation
The most frequent early symptom of hyperparathyroidism is hypercalcemia on normal blood work conducted for other reasons. Most individuals have moderate hypercalcemia, which may be intermittent and is generally less than 1.0 mg/dL over normal upper limits. Unexplained hypercalcemia on a repeat set of test results necessitates additional research.
PTH levels should be checked afterwards, and they may be increased or even within normal ranges, which is inappropriate given the patient's hypercalcemia. Urine calcium levels can be tested every 24 hours, however this is not required for diagnosis.
Radiographic imaging is the next stage in the assessment process. Imaging is critical because it not only informs surgical planning but also distinguishes between a single adenoma and a multi-gland hyperplastic condition.
Because normal parathyroid glands are too tiny to be observed on ultrasonography, the presence of this gland should raise the possibility of a pathogenic condition. On ultrasonography, an adenoma should show as a homogeneously hypoechoic extrathyroidal oval mass with a fat plane separating it from normal thyroid tissue.
A feeding artery may also be seen entering the adenoma's superior or inferior pole. Because it is operator-dependent, ultrasound has a sensitivity of just 60 to 80 percent. It might be used as a supplement to nuclear imaging.
Single-radioisotope scintigraphy with technetium-99m (99mTc) paired with single-photon emission computed tomography (SPECT) imaging is the gold standard for parathyroid localization. SPECT is a three-dimensional sestamibi scan that increases visibility of the parathyroid glands. The combination of these approaches provides a sensitivity of 91 to 98 percent for detecting parathyroid adenomas.
Dual-radioisotope imaging, which uses both 99mTc pertechnetate and thallium-201 (201Tl), is another option. Thallium is taken up by both the thyroid and the parathyroid, whereas pertechnetate is solely taken up by the thyroid. The glands are then seen using subtraction imaging. The need for subtraction software, extended imaging periods, and limited vistas are all disadvantages of this technology.
Four-dimensional computed tomography (CT) and magnetic resonance imaging (MRI) has also been employed in parathyroid imaging. However, they are only 75% sensitive and 40% to 85% sensitive, respectively. As a result, nuclear imaging is used for preoperative localization. Four-dimensional CT and MRI may be more useful in locating ectopic glands following unsuccessful parathyroidectomy.
Management
Traditionally, the surgery of choice for hyperparathyroidism was bilateral 4-gland exploration. However, because a single adenoma accounts for 85 percent of the illness, minimally invasive parathyroidectomy is quickly becoming the gold standard for parathyroid adenoma.
Due to PTH's short half-life, the Miami criterion requires that the PTH level decline by at least 50% within 10 minutes post adenoma excision. If this value is not fulfilled, more investigation is required. Glands that are missing can be located in the retropharyngeal or retro-esophageal planes, the posterior mediastinum, the thymus, the mediastinum, inside the thyroid capsule or gland, or in the carotid sheath.
Complications
Untreated parathyroid adenoma might lead to hypercalcemia consequences. A parathyroid crisis is a rare clinical event characterized by exceptionally high calcium levels, generally greater than 15 mg/dL. Changes in mental state can lead to nervous system breakdown and coma.
Complications from removal of the parathyroid adenoma are also possible. Injury to the recurrent laryngeal nerve is a significant complication of endocrine surgery that can result in hoarseness (unilateral injury) or airway obstruction (bilateral damage). Damage to the nerve can occur as a result of direct trauma such as cutting, grabbing, or stretching. Skeletonization, heat injury, and compression are all examples of indirect nerve damage.
In situations of compression or stretching, the damage may be transitory and resolve in 4 to 6 weeks. If no improvement in function is apparent after 6 to 12 months, the damage should be considered permanent. If the damage is discovered during the procedure, urgent primary repair or grafting should be undertaken.
Primary hyperparathyroidism
Primary hyperparathyroidism is a reasonably common illness that can lead to serious renal and skeletal problems, while most individuals identified in recent decades have modest hypercalcemia and are frequently asymptomatic. Surgery is still the only effective therapy. Conservative observation or medicinal therapy, on the other hand, may be acceptable for certain people.
Etiology
PTH-dependent Causes of Hypercalcemia
- Primary Hyperparathyroidism
- Familial causes of hyperparathyroidism
- Multiple endocrine neoplasia Type 1
- Multiple endocrine neoplasia Type 2
- Familial hyperparathyroidism
- Hyperparathyroidism-jaw tumor syndrome
- Single adenoma
- Multigland disease
- Parathyroid carcinoma
- Familial hypocalciuric hypercalcemia, autosomal dominant inactivating mutations of the calcium-sensing receptor
- An adverse effect of treatment with lithium
PTH Independent Causes of Hypercalcemia
- Malignancy
- Granulomatous diseases
- Hyperthyroidism
- Thiazide therapy
- Vitamin D intoxication
- Milk-alkali syndrome
- Adrenal insufficiency
- Vitamin A intoxication
Epidemiology
Primary hyperparathyroidism, formerly thought to be a rather unusual condition with severe morbidity, is now a relatively common ailment that is frequently asymptomatic. Prior to 1970, the diagnosis went unnoticed until patients complained of particular symptoms, such as nephrolithiasis or bone discomfort.
Since then, most people are diagnosed with hyperparathyroidism when a chemical profile done as a screening test or for an unrelated condition reveals an increase in blood calcium. In the United States, the current incidence is estimated to be 66 per 100,000 person-years in women and 13 to 36 per 100,000 person-years in males.
Pathophysiology
- Normal Calcium Homeostasis
The content of calcium in the extracellular fluid is kept within a fairly restricted range under physiological conditions. Normal calcium homeostasis is based on a complex set of hormonal regulatory processes, including the effects of parathyroid hormone, vitamin D metabolites, and calcitonin on calcium transport in bone, kidney, and GI tract.
About half of total serum calcium is protein-bound, primarily to albumin. 45 percent is ionized, with a little fraction complexed to anions such phosphate and citrate. Despite the fact that only ionized calcium is physiologically active, most labs report total blood calcium levels. Ionized calcium measurements are available. However, by compensating for variances in serum albumin levels, an approximate correction of serum calcium may be produced.
In individuals with hypoalbuminemia, normal total serum calcium levels should be evaluated with caution. These people may have increased ionized calcium levels and be classified as hypercalcemic. When there is a low total calcium concentration in the presence of hypoalbuminemia, the ionized calcium is generally normal.
- Parathyroid Hormone
The concentration of ionized calcium in the extracellular fluid is inversely linked to parathyroid hormone secretion. The calcium-sensing receptor (CaSR) is a G-protein coupled receptor whose activity varies with the kind of calcium in the blood. As the calcium concentration in the extracellular fluid rises, this receptor is activated, and parathyroid cells reduce parathyroid hormone release.
As calcium levels fall, the CaSR activity drops and parathyroid hormone release increases. The etiology of familial hypocalciuric hypercalcemia (FHH) involves mutations that inactivate the CaSR, an autosomal dominant condition characterized by elevated parathyroid hormone production, hypercalcemia, and hypocalciuria.
Thyroid hormone stimulates the parathyroid hormone receptor, increasing calcium and phosphorus resorption from bone, increasing distal tubular calcium resorption, and lowering renal tubular phosphorus resorption. Furthermore, parathyroid hormone is required for vitamin D metabolism by activating the vitamin D 1-alpha hydroxylase, which enhances renal production of 1,25-dihydroxyvitamin D.
Clinical picture
Most patients were diagnosed in previous decades when they complained of nephrolithiasis, bone discomfort, or bone deformities. Most people with primary hyperparathyroidism are now asymptomatic and are identified when hypercalcemia is detected by chance on a chemical profile. Patients should be questioned whether they have a history of kidney stones, bone pain, myalgias or muscular weakness, depressive symptoms, usage of thiazide diuretics, calcium products, vitamin D supplements, or other symptoms linked with the many etiologies of hypercalcemia.
When primary hyperparathyroidism is found at a young age, or there is a family history of hypercalcemia, pituitary adenomas, pancreatic islet cell tumors, pheochromocytomas, or medullary thyroid cancer, a familial condition should be examined.
A patient with primary hyperparathyroidism generally has a normal physical examination. The physical examination, on the other hand, can be useful in detecting anomalies that may indicate other causes of hypercalcemia. Physical examination seldom reveals parathyroid adenomas, but the appearance of a big, hard mass in the neck of a patient with hypercalcemia should raise the possibility of a parathyroid cancer.
Evaluation
Patients with primary hyperparathyroidism and other causes of PTH-dependent hypercalcemia frequently have PTH levels that are clearly high, while others have readings that are within the reference range for the general population. In the context of hypercalcemia, a normal PTH is regarded as inappropriate and is nevertheless consistent with PTH-dependent hypercalcemia. PTH levels in individuals with PTH-independent hypercalcemia should be very low.
In a patient with chronic hypercalcemia and an increased serum level of parathyroid hormone, a full clinical assessment supplemented by regular laboratory and radiologic investigations should be adequate to establish a diagnosis of primary hyperparathyroidism. Hypercalcemia caused by clinically occult tumors is rare. The majority of individuals with malignancy-associated hypercalcemia have cancer or have cancer that is easily apparent on first examination, and their PTH levels will be decreased.
A study of past medical records can frequently be very helpful in determining the etiology of hypercalcemia. Most hyperparathyroid patients have chronic or intermittent hypercalcemia for several years before a clear diagnosis is made. Other than hyperparathyroidism, few disorders can allow a healthy-appearing individual to be hypercalcemic for more than a few years without becoming clinically noticeable.
List of tests for primary hyperparathyroidism:
- Total calcium
- Albumin
- Ionized calcium in selected cases when there are questions about the accuracy of the corrected calcium
- Parathyroid hormone
- Phosphorus
- BUN and creatinine
- Alkaline phosphatase
- 25-hydroxyvitamin D
- Urine calcium and creatinine
- Imaging to screen for renal calcifications or urolithiasis
- Bone densitometry (DXA) including measurement at the distal 1/3 radius
- EKG
- Genetic testing in selected individuals if there is suspicion of a genetic syndrome
- A parathyroid scan and a neck ultrasound are both recommended. Because of the possibility of false negative results, these tests are not considered diagnostic. They should not be ordered if surgery is not planned. They should be prescribed when surgery is planned to aid the surgeon in localizing an enlarged parathyroid gland.
Management
Although surgery is still the ultimate treatment for primary hyperparathyroidism, non-operative monitoring may be acceptable for some individuals, notably the elderly with modest hypercalcemia and no severe sequelae. In certain people, medical therapy with bisphosphonates or cinacalcet may be beneficial. The decision to suggest surgery is based on the patient's age, the degree of hypercalcemia, and the existence or absence of hyperparathyroidism-related comorbidities. For patients who have recurring kidney stones, surgery is the therapy of choice.
Several workshops have been held since 1990 to produce guidelines to help clinicians treat asymptomatic hyperparathyroidism. Internationally acknowledged surgical and medical professionals with expertise in handling patients with hyperparathyroidism evaluated the evidence-based medical literature, and a consensus of their viewpoints was conveyed to the medical community. The most current set of recommendations was issued in 2014.
The current guidelines state that surgery should be recommended for asymptomatic primary hyperparathyroidism when:
- Serum calcium is more than 1 mg/dL greater than the upper limit of normal
- Age younger than 50 years
- Osteoporosis
- GFR less than 60 mL/min
- Urine calcium greater than 400 mg/24 hours
- Evidence of renal calcification or stones
Untreated, many individuals with primary hyperparathyroidism experience gradual loss of cortical bone, whereas successful treatment results in a significant increase in bone mineral density, which can last for up to 15 years. If observation is the chosen course of action, patients must be monitored on a regular basis with measurements of blood and urine calcium, renal function, and bone densitometry. If hypercalcemia worsens or problems emerge, surgery should be considered.
- Medical Treatment
Some individuals with primary hyperparathyroidism who are not surgical candidates may benefit from medicinal therapy.
- Bisphosphonates can increase bone mineral density in those with osteoporosis or osteopenia.
- Agonists to the calcium-sensing receptor, such as cinacalcet will lower PTH and calcium levels. However, they do not increase bone density
Parathyroid Cancer
HPT can be caused by parathyroid carcinoma. This extremely uncommon condition mainly affects persons in their 50s. After therapy, it frequently returns or recurs at the original location. Controlling the level of calcium in the blood can help to delay recurrence.
Early detection is critical for survival. Hypercalcemia and its repercussions are frequently more dangerous to your health than cancer itself.
Hypoparathyroidism
Hypoparathyroidism occurs when the body does not produce enough PTH. As a result, the calcium level in the blood is abnormally low. The illness is frequently caused by neck surgery or parathyroid injury. It can also occur as a result of autoimmune assaults on the glands.
Hypoparathyroidism increases the risk of:
- Addison's disease is a condition in which the adrenal glands fail to produce adequate hormones. Only autoimmune-related hypoparathyroidism is connected with this risk.
- Cataracts.
- Parkinson's disease, is a progressive disorder that affects the brain and causes abnormal movements and other symptoms.
- Pernicious anemia is a disorder in which the body is unable to produce enough red blood cells owing to a vitamin B-12 deficiency. People suffering with this illness are unable to absorb enough B-12 from their meals. Only autoimmune-related hypoparathyroidism is connected with this risk.
Conclusion
The parathyroids, four pea-sized glands found in the neck near the thyroid, are affected by parathyroid illness. Parathyroid hormone is produced by the glands (PTH). PTH aids in the maintenance of the proper calcium balance in the body. Normally, the glands release just enough PTH to maintain appropriate calcium levels. This delicate equilibrium is thrown off by disease.
Primary hyperparathyroidism lowers cortical bone density and increases fracture risk in areas where cortical bone predominates, such as the distal forearm while protecting trabecular bone. As a result, those with hyperparathyroidism should have dual-energy x-ray absorptiometry that includes the distal third radius, which is virtually entirely made of cortical bone, as well as measures at the spine and hip.
Depression, anxiety, weariness, irritability, lassitude, and sleep disruption are some of the psychological symptoms linked with primary hyperparathyroidism. Following parathyroidectomy, there is uneven improvement in psychological indices and quality of life, and patient numbers in high-quality research are modest.