Pulmonary embolism (PE)
Overview
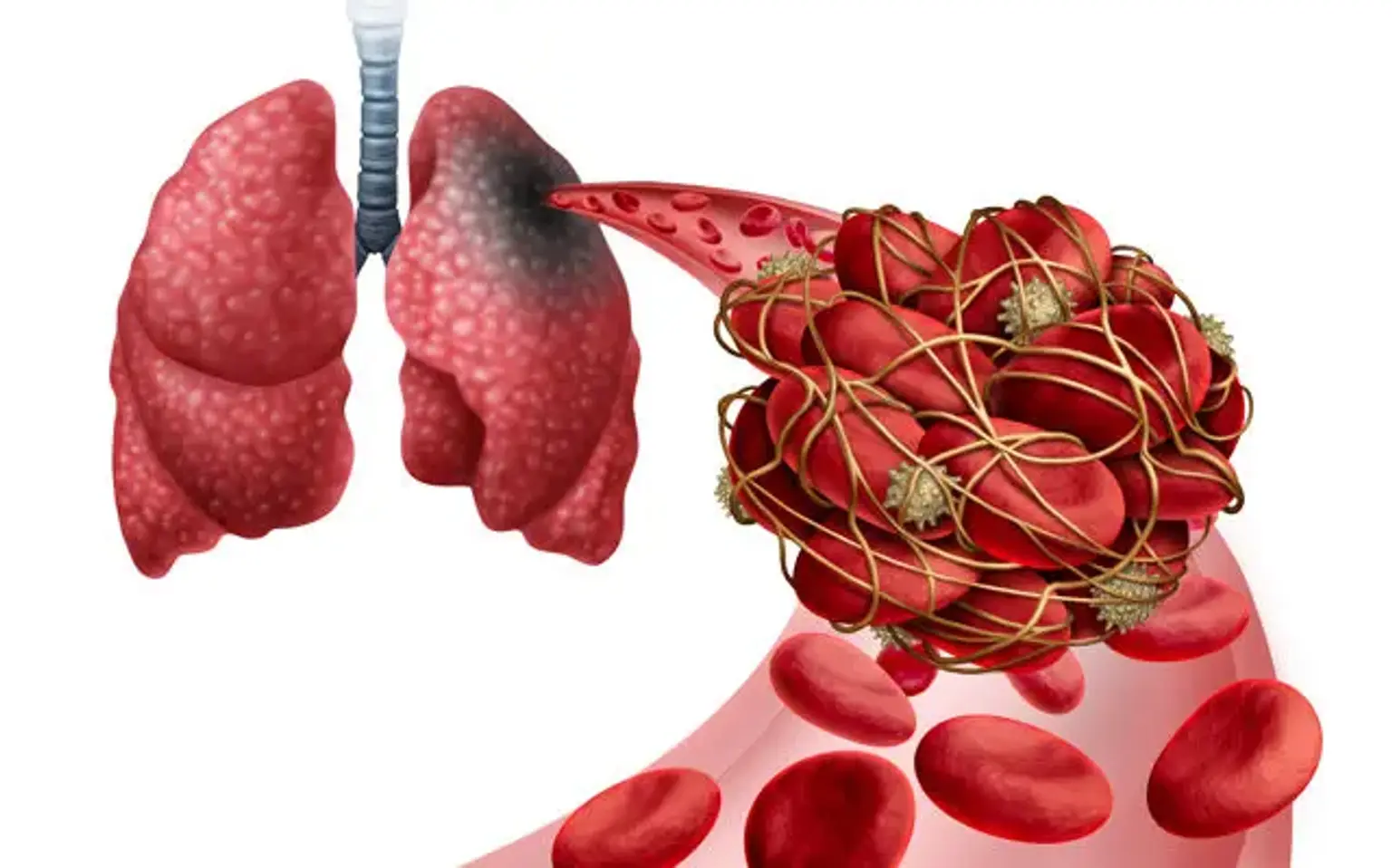
When a thrombus from someplace else disrupts the flow of blood in the pulmonary artery or its branches, this is referred to as pulmonary embolism (PE). A thrombus forms within the deep veins, most often in the lower limbs, in deep vein thrombosis (DVT). PE is often caused when a portion of the thrombus breaks off and enters the pulmonary circulation.
PE can develop very infrequently as a result of embolization of various materials into the pulmonary circulation, such as air, fat, or tumor cells. The combined spectrum of PE and DVT is known as venous thromboembolism (VTE).
What is a pulmonary embolism?
A pulmonary embolism (PE) occurs when a blood clot (thrombus) gets lodged in a lung artery, obstructing blood flow to the lung. Pulmonary embolism is generally caused by a thrombus that forms in the deep venous system of the lower extremities. With a wide spectrum of clinical manifestations ranging from asymptomatic to fatal, diagnosing PE can be difficult.
There are a variety of tools available, including as clinical grading systems, laboratory data, and imaging investigations, to assist doctors in their work-up of PE. Early detection and treatment are critical for reducing the mortality and morbidity associated with PE.
Epidemiology
The yearly incidence of pulmonary embolism (PE) varies from 39 to 115 per 100,000 persons, whereas that of deep vein thrombosis (DVT) ranges from 53 to 162 per 100,000 people. Acute pulmonary embolism is the third most prevalent kind of cardiovascular illness, behind coronary artery disease and stroke. Males are shown to have a higher incidence of PE than females. Overall, PE-related mortality is significant, with 100,000 fatalities occurring each year in the United States.
However, estimating the fatality rates related to PE can be difficult because many individuals with sudden cardiac death are assumed to have experienced a thromboembolic event such as PE. It is crucial to note that the case-fatality rates of PE have been lowering; this might be due to advancements in diagnostic methods and the implementation of early intervention and therapy.
Pulmonary embolism causes
The majority of pulmonary embolisms begin as lower extremity DVTs. As a result, risk factors for pulmonary embolism (PE) are the same as risk factors for deep vein thrombosis (DVT). Understanding these risk variables is provided by Virchow's triad of hypercoagulability, venous stasis, and endothelial damage.
There are two types of risk factors: hereditary and acquired. Thrombophilia, such as factor V Leiden mutation, prothrombin gene mutation, protein C deficiency, protein S deficiency, and hyperhomocysteinemia are all genetic risk factors.
Immobilization for an extended period of time (bed rest for more than three days, anyone traveling for more than four hours by air, car, bus, or train), recent orthopedic surgery, malignancy, indwelling venous catheter, obesity, pregnancy, cigarette smoking, oral contraceptive pill use, and so on are all acquired risk factors.
Other predisposing factors for VTE include:
- Fracture of lower limb
- Hospitalization for heart failure or atrial fibrillation/flutter within the previous three months
- Hip or knee replacement
- Major trauma
- History of previous venous thromboembolism
- Central venous lines
- Chemotherapy
- Congestive heart failure or respiratory failure
- Hormone replacement therapy
- Oral contraceptive therapy
- Postpartum period
- Infection (specifically pneumonia, urinary tract infection, and HIV)
- Cancer (highest risk in metastatic disease)
- Thrombophilia
- Bed rest greater than three days
- Obesity
- Pregnancy
Cancer increases the likelihood of thrombus development and, as a result, PE. VTE is most common in pancreatic cancer, hematological malignancies, lung cancer, gastric cancer, and brain cancer. Infection in any part of the body is a prevalent cause of VTE. PE is made more likely by myocardial infarction and congestive heart failure (CHF). Patients with VTE were also shown to have a higher risk of future stroke and myocardial infarction.
Types of Pulmonary Embolism
It is critical to categorize PE depending on the presence or lack of hemodynamic stability.
Hemodynamically unstable PE (previously known as massive or high-risk PE) is defined as PE that causes hypotension (as defined by systolic blood pressure (SBP) less than 90 mmHg or a drop in SBP of 40 mmHg or more from baseline or hypotension that requires vasopressors or inotropes). The term "massive" PE does not describe the size of the PE but rather its hemodynamic effect. Patients with hemodynamically unstable PE have a higher risk of dying from obstructive shock (i.e., severe right ventricular failure).
Hemodynamically stable PE encompasses a spectrum of conditions ranging from small, mildly symptomatic, or asymptomatic PE (low-risk PE or small PE) to PEs that cause mild hypotension that stabilizes in response to fluid therapy or those that present with right ventricle dysfunction (submassive or intermediate-risk PE) but are hemodynamically stable.
Pathophysiology
When clots break off and embolize into the pulmonary circulation, this is referred to as pulmonary embolism. Pulmonary emboli are usually many, with the lower lobes more commonly affected than the higher, and bilateral lung involvement more prevalent.
Large emboli have a tendency to clog the major pulmonary artery, resulting in saddle embolus, which has negative cardiovascular implications. Smaller emboli, on the other hand, can block peripheral arteries and cause pulmonary infarction, as seen by intra-alveolar bleeding. In roughly 10% of patients, a pulmonary infarction develops.
PE impairs gas exchange owing to pulmonary vascular bed blockage, resulting in a mismatch in the ventilation to perfusion ratio because alveolar ventilation remains constant while pulmonary capillary blood flow drops, thereby resulting in dead space ventilation and hypoxemia.
In addition, mediators such as serotonin are produced, resulting in vasospasm and reduced pulmonary flow in unaffected parts of the lung. Local inflammatory mediator buildup changes lung surfactant and promotes respiratory drive, resulting in hypocapnia and respiratory alkalosis.
The mechanical blockage of the arterial bed with thrombus and hypoxic vasoconstriction raises pulmonary vascular resistance (PVR) in PE. If thromboemboli occlude more than 30% to 50% of the entire cross-sectional area of the pulmonary arterial bed, pulmonary artery pressure (PAP) rises.
Increased PVR causes right ventricular afterload to rise, obstructing right ventricular outflow and causing right ventricular dilatation and flattening or bending of the interventricular septum. The development of the right bundle branch block may exacerbate ventricle desynchronization. Reduced RV outflow and accompanying RV dilation diminish left ventricular filling, putting cardiac output at risk.
As a result, LV filling is decreased in early diastole, resulting in a decrease in cardiac output (CO), systemic hypotension, and hemodynamic instability. The leading cause of mortality in severe PE is right ventricular (RV) failure owing to abrupt pressure overload. Given the aforementioned pathophysiological reasons, clinical symptoms, evidence of overt RV failure, and hemodynamic instability all point to a substantial likelihood of early (in-hospital or 30 day) death.
Symptoms of pulmonary embolism
A quick identification of a pulmonary embolism (PE) is critical due to the high associated mortality and morbidity, both of which can be avoided with early treatment. It is vital to highlight that 30% of untreated individuals with pulmonary embolism die, but only 8% die following early treatment. Unfortunately, because to the large variety of nonspecific clinical signs and symptoms in individuals with acute PE, the diagnosis of PE can be problematic.
Dyspnea, pleuritic chest discomfort, cough, hemoptysis, presyncope, or syncope are the most prevalent signs of PE. Dyspnea in central PE can be acute and severe, but it is usually moderate and temporary in modest peripheral PE. Worsening dyspnea may be the only symptom in people who already have heart failure or pulmonary illness.
Chest discomfort is a common symptom induced by pleural irritation produced by distant emboli causing pulmonary infarction. Chest discomfort in central PE may be caused by underlying right ventricular (RV) ischemia and must be distinguished from an acute coronary syndrome or aortic dissection.
Arrhythmias (e.g., atrial fibrillation), syncope, and hemodynamic collapse are less common presentations. Hemodynamic instability is an uncommon but important clinical manifestation since it suggests central or widespread PE with substantially diminished hemodynamic reserve. Syncope can develop and is linked to an increased risk of hemodynamic instability and RV dysfunction. It is critical to realize that people with big PE may be asymptomatic or have minimal symptoms at times. PE is frequently asymptomatic or identified by chance during a diagnostic workup for another condition.
Patients with PE may show tachypnea and tachycardia on examination, which are frequent but nonspecific symptoms. Other exam findings may include calf swelling, tenderness, erythema, palpable cords, pedal edema, rales, decreased breath sounds, signs of pulmonary hypertension such as elevated neck veins, a loud P2 component of the second heart sound, a right-sided gallop, and a right ventricular parasternal lift.
PE is a well-known cause of sudden cardiac death (8 percent ). A major PE causes immediate right ventricular failure, characterized by jugular venous distension, parasternal lift, third heart sound, cyanosis, and shock. If a patient with PE who had tachycardia at the time of presentation develops rapid bradycardia or a new wide complex tachycardia (with right bundle branch block), clinicians should search for symptoms of right ventricular strain and potential approaching shock. Anyone who has hypotension with jugular venous distension and has ruled out acute myocardial infarction, pericardial tamponade, or tension pneumothorax should be suspected of having PE.
Diagnosis
Diagnostic Workup
Arterial Blood Gas (ABG) Analysis
Unexplained hypoxemia in the presence of a normal chest radiograph should raise the clinical suspicion for pulmonary embolism (PE). ABG results such as widened alveolar-arterial gradient for oxygen, respiratory alkalosis, and hypocapnia are typical as a pathophysiological response to pulmonary embolism. It should be noted that while hypercapnia, respiratory or lactic acidosis are uncommon, they can occur in individuals with major PE accompanied with obstructive shock and respiratory arrest.
Brain Natriuretic Peptide (BNP)
In individuals suspected of having PE, elevated BNP offers little diagnostic use. Acute PE causes right ventricular pressure overload, which causes myocardial strain and the release of B-type natriuretic peptide (BNP) and N-terminal (NT)-proBNP. Thus, natriuretic peptide levels in the blood represent the degree of RV dysfunction in acute PE.
Troponin
Serum troponin I and T levels are advantageous in terms of prognosis but not in terms of diagnosis. Troponin levels are raised in 30 to 50 percent of individuals with moderate to large PE and are connected to clinical worsening and mortality following PE as indications of right ventricular dysfunction.
D-dimer
D-dimer levels in plasma rise when there is an acute thrombotic process in the body due to the simultaneous activation of coagulation and fibrinolysis pathways. Because D-dimer testing has a strong negative predictive value, a normal D-dimer result indicates that acute PE or DVT is unlikely. However, because increased D-dimer levels have a low positive predictive value, D-dimer testing is ineffective for PE confirmation.
Because there are several D-dimer assays available, physicians should get familiar with the diagnostic performance of the test utilized in their clinical environment. The quantitative enzyme-linked immunosorbent test (ELISA) has at least 95% diagnostic sensitivity. It can be utilized to rule out PE in individuals with a low or moderate pretest likelihood. A negative ELISA D-dimer test, along with a low clinical likelihood, can rule out PE in around 30% of suspected patients without additional testing.
The specificity of D-dimer declines rapidly with age, reaching around 10% in individuals over the age of 80. The adoption of age-adjusted cut-offs for patients over the age of 50 may enhance D-dimer testing results in the elderly. In one research, using the age-adjusted cut-off instead of the usual D-dimer cut-off of 500 ng/mL or above raised the proportion of patients in whom PE could be ruled out from 6.4 percent to 30 percent, with no extra false-negative results.
Electrocardiography (ECG)
In individuals with suspected PE, ECG abnormalities are vague. Tachycardia and nonspecific ST-segment and T-wave alterations are the most prevalent ECG findings in PE. S1Q3T3 pattern, right ventricular strain, and new incomplete right bundle branch block are unusual.
Chest Radiograph (CXR)
CXR is normally normal in PE, although it may indicate nonspecific abnormalities such as atelectasis or effusion. It aids in ruling out alternate diagnosis in individuals with acute dyspnea.
Computed Tomographic Pulmonary Angiography (CTPA)
For patients with suspected PE, multidetector CTPA is the preferred diagnostic method. A normal CTPA demonstrated a strong negative predictive value for PE of 96 percent and 89 percent in patients with a low or intermediate clinical likelihood, respectively, but only 60 percent in individuals with a high pretest probability.
Pulmonary Angiography
In pulmonary angiography, contrast is injected into the right heart using a catheter under fluoroscopy, which was once the gold standard for the diagnosis of PE. Acute PE is diagnosed based on the presence of a thrombus, which can be seen as amputation of a pulmonary artery branch or a filling defect. With the widespread use of CTPA, pulmonary angiography is now only done in rare cases for individuals with a high clinical likelihood of PE for whom CTPA or V/Q screening is nondiagnostic.
The outcomes of pulmonary angiography appear to be inferior to CTPA, and they are operator dependent and very variable. As a result, catheter-based pulmonary angiography is performed in patients who require therapeutic benefit since it aids in both diagnostic and therapeutic measures focused at clot lysis.
Magnetic Resonance Angiography
For some years, magnetic resonance angiography (MRA) has been evaluated in the context of suspected PE. Large-scale studies, however, demonstrate that, while promising, this approach is not recommended as a first-line diagnostic for the diagnosis of PE because to its low sensitivity, limited availability in most emergency settings, and high number of inconclusive MRA scans.
Echocardiography
When a thrombus is seen in the proximal pulmonary arteries, transthoracic echocardiography can be used to make a conclusive diagnosis of PE. The presence of a clot in the right heart or new right heart strain supports the diagnosis of PE on echocardiography, especially in hemodynamically unstable patients with suspected PE, where an echocardiogram may be useful to establish a possible diagnosis and justify the emergency use of thrombolytic therapy.
There are several factors to consider while utilizing echocardiography to diagnose PE. Because of the RV's unique structure, there is no one echocardiographic measure that provides rapid and precise information on RV size or function. As a result, the echocardiographic criteria for the diagnosis of PE have differed between investigations.
Compression Ultrasonography (US)
In the vast majority of cases, PE is caused by a lower-limb DVT, and very rarely by an upper-limb DVT (mostly following venous catheterization). DVT was discovered in 70% of individuals with confirmed PE in one investigation. For proximal symptomatic DVT, Compression US has a sensitivity of more than 90% and a specificity of roughly 95%. In individuals suspected of having PE, a detection of proximal DVT is considered sufficient to warrant anticoagulant medication without additional testing.
Because of its limited sensitivity, compression ultrasonography is reserved for individuals for whom definitive imaging (e.g., CTPA, V/Q scanning) is contraindicated or inconclusive.
The Wells criterion and the Geneva score are the most often used scoring systems to determine the pretest chance of having a PE. This enables the categorization of patients with suspected PE into clinical or pretest likelihood groups, from which diagnostic tests are chosen and interpreted.
Wells Criteria and Modified Wells Criteria
Items/Scores
- Clinical symptoms of DVT-3.0
- Other diagnoses less likely than pulmonary embolism-3.0
- Heart rate >100 beats per min-1.5
- Immobilization for three or more days or surgery in the previous four weeks-1.5
- Previous history of DVT-PE1.5
- Hemoptysis-1.0
- Malignancy-1.0
Probability/ Score
Traditional Clinical Probability Assessment (Wells criteria)
- High/>6.0
- Moderate/2.0 to 6.0
- Low/<2.0
Simplified Clinical Probability Assessment (Modified Wells Criteria)
- PE likely/>4.0
- PE unlikely/≤4.0
Pulmonary embolism treatments
A) Initial Management
1. The Supportive Measures
The first line of treatment for individuals with pulmonary embolism (PE) should be supportive measures.
Supplemental oxygen is recommended for patients with an oxygen saturation of 90% or higher. In unstable patients, mechanical ventilation (non-invasive or invasive) should be used, but physicians should be aware of the negative hemodynamic implications of mechanical ventilation.
In patients with hemodynamically unstable PE, acute RV failure is the primary cause of mortality. In such cases, aggressive volume resuscitation might overstrain the RV, aggravate ventricular interdependence, and limit cardiac output (CO). As a result, only individuals with collapsing IVC/intravascular depletion should be considered for intravenous fluid resuscitation in patients with significant PE. Hemodynamic support may need the use of vasopressors.
In hemodynamically unstable patients with pulmonary embolism, mechanical cardiopulmonary support devices, such as extracorporeal membrane oxygenation (ECMO), may be employed.
2. Anticoagulation
It is critical to remember that anticoagulation is the cornerstone of acute PE therapy.
It is vital to highlight that anticoagulation in acute PE can be achieved with either low-molecular-weight heparin (LMWH), fondaparinux, or unfractionated heparin (UFH). Because they are less likely to cause significant bleeding and heparin-induced thrombocytopenia, LMWH and fondaparinux are chosen. UFH is often utilized exclusively in patients with hemodynamic instability who may require primary reperfusion therapy, or in patients with renal impairment. Anticoagulation in PE can also be achieved with the use of newer oral anticoagulants (NOACs) and vitamin K antagonists (VKA).
Treatment for individuals with suspected PE is stratified based on the kind of PE (whether hemodynamically stable or unstable PE) and the particular patient's suspicion of PE. Based on the updated Geneva or Wells score, patients are classed as having a low, middle, or high suspicion for PE.
- Hemodynamically Stable Patients:
Anticoagulation is begun in patients with a strong clinical suspicion of PE even before diagnostic imaging is acquired. If diagnostic imaging can be done within 24 hours for individuals with a low clinical suspicion for PE, then wait for imaging to confirm a definite diagnosis before initiating anticoagulant medication.
If diagnostic imaging can be done within 4 hours for individuals with an intermediate clinical suspicion for PE, then wait for imaging to confirm a definite diagnosis before initiating anticoagulant medication.
Once the diagnosis of PE is confirmed, IVC filter installation should be explored for individuals for whom anticoagulation is contraindicated.
- Hemodynamically Unstable Patients:
When feasible, patients with a strong clinical suspicion for PE who are hemodynamically unstable should have urgent CTPA, portable perfusion scanning, or bedside transthoracic echocardiography done. For patients with hemodynamically unstable acute PE, primary reperfusion therapy, mainly thrombolysis, is the treatment of choice.
In individuals who cannot have thrombolysis, surgical pulmonary embolectomy or percutaneous catheter-directed treatment are possibilities for reperfusion. Patients recovering from high-risk PE can be transferred from parenteral to oral anticoagulation after reperfusion therapy and hemodynamic stability.
3. Reperfusion Strategies
- Thrombolysis:
When compared to UFH alone, thrombolysis has demonstrated an effective reduction in pulmonary artery pressure and resistance in individuals with PE; these benefits are measured by a decrease in RV dilation on echocardiography. Thrombolysis is favored when therapy can be started within 48 hours of symptom onset, although it has also been demonstrated to help individuals whose symptoms started less than 14 days earlier.
The Pulmonary Embolism Thrombolysis (PEITHO) study found that thrombolysis was beneficial in hemodynamically stable individuals with intermediate-risk PE. It found that thrombolysis was related with a considerable reduction in the risk of hemodynamic decompensation or collapse, but it also found that thrombolytics increased the risk of serious bleeding.
Any prior intracranial hemorrhage, known structural intracranial cerebrovascular disease (e.g., arteriovenous malformation), known malignant intracranial neoplasm, ischemic stroke within three months, suspected aortic dissection, active bleeding or bleeding diathesis, recent surgery encroaching on the spinal canal or brain, and recent significant closed-head or facial trauma with radiographic evidence of bony fracture or fracture diathesis are absolute contraindications to thro
- Catheter-Directed Treatment:
Involves inserting a catheter into the pulmonary arteries and using it for ultrasound-assisted thrombolysis, suction embolectomy, rotational embolectomy, thrombus aspiration, or combining mechanical fragmentation with pharmaceutical catheter-directed thrombolysis. Catheter-directed therapy have been proven in trials to have a success rate of up to 87 percent. Catheter-assisted embolectomy procedures include the inherent danger of perforating the pulmonary arteries, which might result in significant hemoptysis or cardiac tamponade. These problems are uncommon yet deadly.
- Surgical Embolectomy:
It is often suggested in patients with hemodynamically unstable PE who are contraindicated for thrombolysis (systemic or catheter-directed), or in patients who have failed thrombolysis. There was no difference in mortality between thrombolysis and surgical embolectomy, but the thrombolysis group had a greater risk of stroke and re-intervention.
- Vena Cava Filters:
These obstruct emboli's course of travel and keep them from entering the pulmonary circulation. Filters are recommended in individuals with venous thromboembolism who are anticoagulant-intolerant and in those who have recurrent VTE despite anticoagulation. Retrievable filters are desired, so that once the contraindication has been addressed, the filter may be withdrawn and the patient can be anticoagulated.
This is due to the Prevention of Recurrent Pulmonary Embolism by Vena Cava Interruption (PREPIC) study, which found that inserting a permanent vena cava filter was associated with a significant reduction in the risk of recurrent PE and a significant increase in the risk of DVT, but no significant difference in the risk of recurrent VTE or death.
B. Chronic Treatment and Prevention of Recurrence
The goal of anticoagulation following acute PE therapy is to finish the acute phase while also preventing VTE recurrence in the long run. Various durations of anticoagulant treatment with vitamin K antagonists (VKAs) for VTE have been studied in clinical studies.
The findings of these investigations have led to the following conclusions. To begin, all patients with PE should be treated with anticoagulants for at least three months. Second, when anticoagulant medication is discontinued, the risk of recurrence is likely to be comparable if anticoagulants are discontinued after 3-6 months vs longer treatment durations (e.g., 12-24 months).
Third, extended oral anticoagulant therapy decreases the risk of recurrent VTE by 90%, although the danger of bleeding balances this benefit slightly. Oral anticoagulants are extremely effective in avoiding recurrent VTE during therapy, but they do not remove the chance of future recurrence when treatment is stopped. It is vital to know that around 30% of PEs are unprovoked.
When compared to individuals who had a provoked PE, unprovoked PE (PE in the absence of a recognized risk factor) is linked with a two- to three-fold increase in the probability of recurrence. Patients with long-term risk factors (such as cancer or increased antiphospholipid antibodies) had a greater recurrence incidence than those with temporary risk factors (e.g., immobilization, surgery, or trauma).
Finally, the ideal duration of anticoagulation remains unknown and must be assessed on a case-by-case basis. A minimum of three months is normally suggested, but if the PE was unprovoked or if there are persisting risk factors, a longer duration is necessary. The necessity for extended anticoagulation should be evaluated at the end of three months, taking into account the patient's bleeding risk. Those who are at high risk of bleeding should restrict their therapy to three months.
Conclusion
Pulmonary embolism (PE) is caused by emboli that move from venous thrombi to the lungs and occluding the arteries. PE is the most serious kind of venous thromboembolism, and it may be deadly if left undetected or untreated. Acute PE is linked to right ventricular dysfunction, which can result in arrhythmia, hemodynamic collapse, and shock.
Individuals who survive PE may also develop post-PE syndrome, which is characterized by chronic thrombotic remnants in the pulmonary arteries, persistent right ventricular dysfunction, poor quality of life, and/or chronic functional restrictions. In recent years, several significant advances in the diagnostic and therapeutic management of acute PE have been made, including the introduction of a simplified diagnostic algorithm for suspected PE and phase III trials demonstrating the value of direct oral anticoagulants in acute and extended treatment of venous thromboembolism.
Future research should focus on innovative therapeutic alternatives (such as fibrinolysis enhancers) as well as improved approaches for predicting long-term problems and determining appropriate anticoagulant medication settings in individual patients.