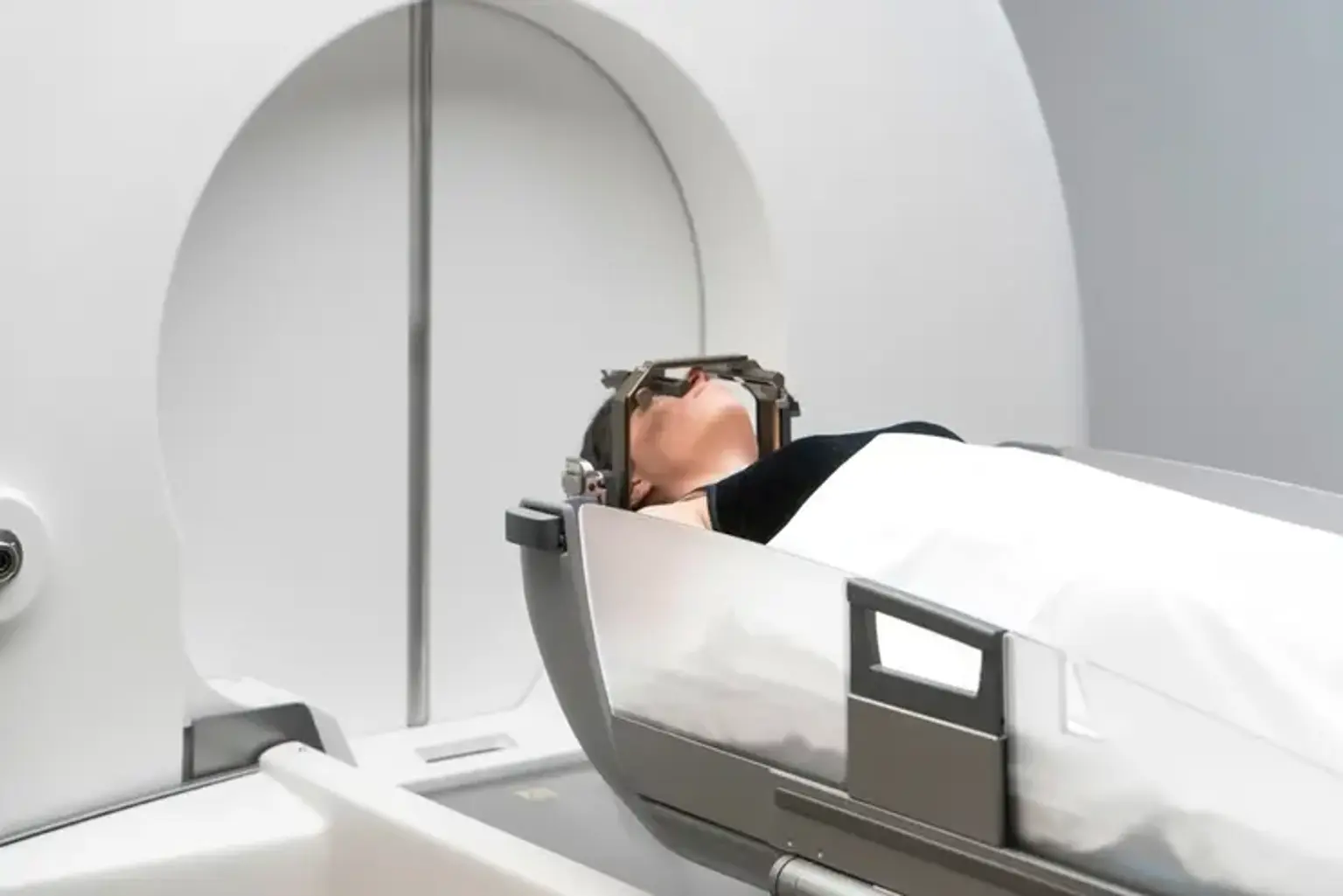
Stereotactic Radiosurgery (SRS)
Stereotactic radiosurgery (SRS) is non-surgical radiotherapy used as a treatment of functional disorders and tiny tumors of the brain. In comparison to conventional therapy, it can administer precisely focused radiation in fewer high-dose treatments, helping to maintain healthy tissue. Stereotactic body radiotherapy (SBRT) is the term used when SRS is utilized to treat body cancers.
Typically, SRS and SBRT are done as outpatient procedures. Ask your doctor if you should arrange for a ride home after the procedure and whether you should skip meals, beverages, or medication several hours before the procedure. If you think you might be pregnant, if you're nursing a baby, or if you use oral diabetic medications or insulin, let your doctor know. Discuss any medical implants, claustrophobia, or contrast material sensitivities you may have.