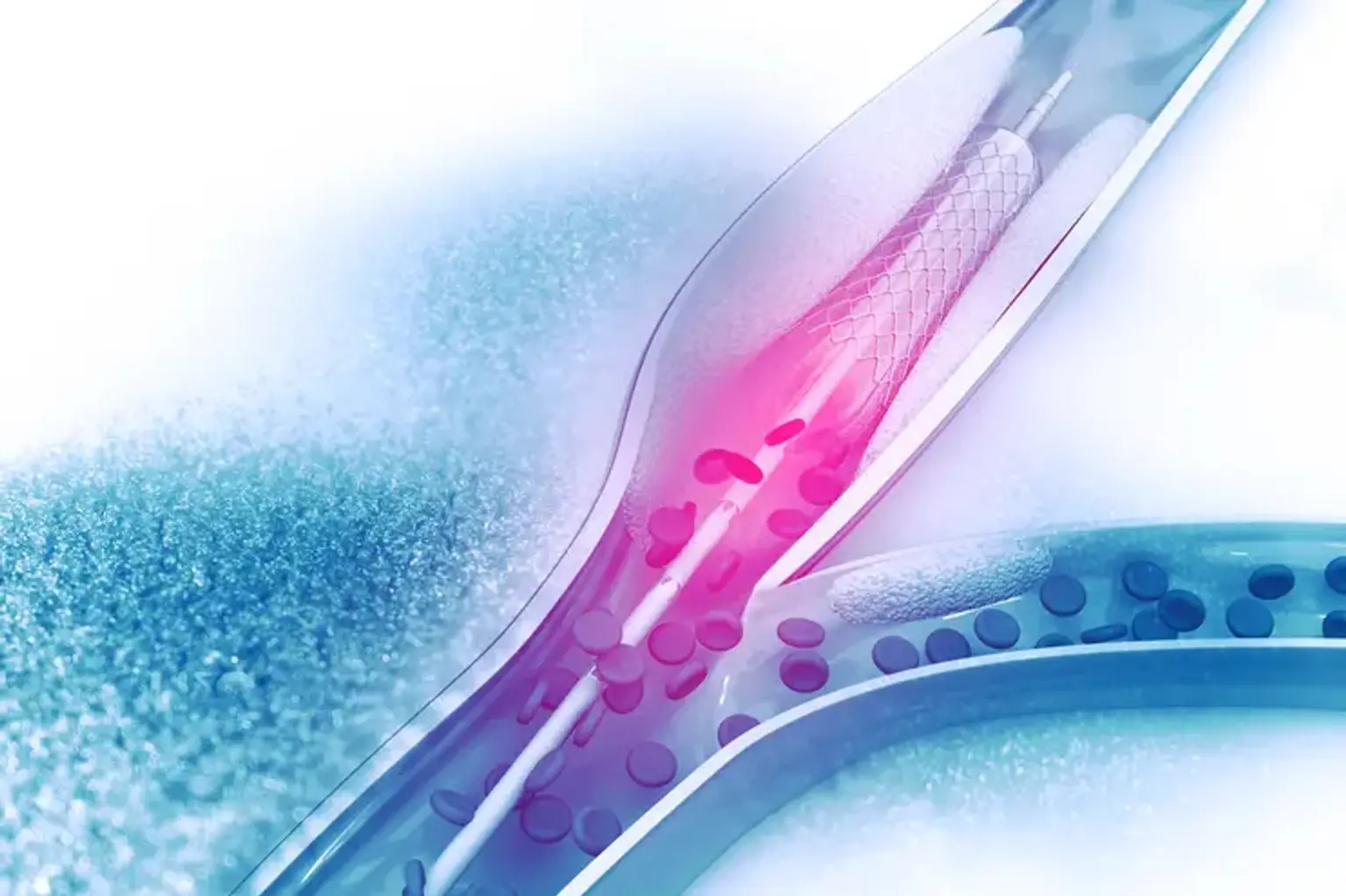
Coronary Angioplasty
Overview
Coronary heart disease (CHD) is common among the elderly globally. 15.5 million individuals in the United States have CHD According to the American Heart Association. It is a substantial cause of mortality and morbidity in industrialized nations, accounting for almost one-third of all deaths in persons over the age of 35.
Because to prompt percutaneous coronary intervention with stenting, CHD mortality has gradually decreased over the previous few decades. As a result, angioplasty is a game changer in terms of lowering morbidity and mortality. For acute myocardial infarction, angioplasty is the preferred therapy. There are two primary catheterization approaches: traditional and transfemoral.